NPs Basic Information

|
Name |
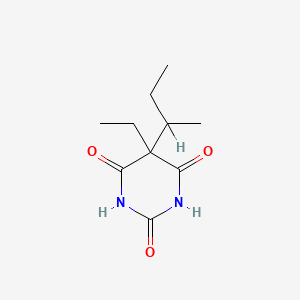
Valylvaline anhydride
|
| Molecular Formula | C10H18N2O2 | |
| IUPAC Name* |
3,6-di(propan-2-yl)piperazine-2,5-dione
|
|
| SMILES |
CC(C)C1C(=O)NC(C(=O)N1)C(C)C
|
|
| InChI |
InChI=1S/C10H18N2O2/c1-5(2)7-9(13)12-8(6(3)4)10(14)11-7/h5-8H,1-4H3,(H,11,14)(H,12,13)
|
|
| InChIKey |
QGMAWEIDGADSAC-UHFFFAOYSA-N
|
|
| Synonyms |
Valylvaline anhydride; 5625-44-5; 3,6-di(propan-2-yl)piperazine-2,5-dione; 3,6-Diisopropyl-2,5-piperazinedione; Cyclo(L-Val-L-Val); L-Valyl-valine anhydride; 2,5-Piperazinedione, 3,6-bis(1-methylethyl)-; SCHEMBL6439632; DTXSID00971680; 3,6-Diisopropylpiperazin-2,5-dione; 3,6-diisopropylpiperazine-2,5-dione; AKOS006281827; 3,6-diisopropylpipera-zine-2,5-dione; 3,6-Diisopropyl-2,5-piperazinedione #; (l,l)-cis-3,6-Diisopropylpiperazine-2,5-dione; 3,6-Di(propan-2-yl)-3,6-dihydropyrazine-2,5-diol; 2,5 Piperazinedione, 3,6-bis(1-methylethyl)-, (35)-trans-
|
|
| CAS | 5625-44-5 | |
| PubChem CID | 519728 | |
| ChEMBL ID | NA |
*Note: the IUPAC Name was collected from PubChem.
Chemical Classification: |
|
|
|---|
——————————————————————————————————————————
NPs Physi-Chem Properties
| Molecular Weight: | 198.26 | ALogp: | 1.4 |
| HBD: | 2 | HBA: | 2 |
| Rotatable Bonds: | 2 | Lipinski's rule of five: | Accepted |
| Polar Surface Area: | 58.2 | Aromatic Rings: | 1 |
| Heavy Atoms: | 14 | QED Weighted: | 0.685 |
——————————————————————————————————————————
NPs ADMET Properties*
ADMET: Absorption
| Caco-2 Permeability: | -4.856 | MDCK Permeability: | 0.00000673 |
| Pgp-inhibitor: | 0 | Pgp-substrate: | 0.37 |
| Human Intestinal Absorption (HIA): | 0.016 | 20% Bioavailability (F20%): | 0.082 |
| 30% Bioavailability (F30%): | 0.063 |
——————————————————————————————————————————
ADMET: Distribution
| Blood-Brain-Barrier Penetration (BBB): | 0.932 | Plasma Protein Binding (PPB): | 41.67% |
| Volume Distribution (VD): | 0.786 | Fu: | 52.72% |
——————————————————————————————————————————
ADMET: Metabolism
| CYP1A2-inhibitor: | 0.018 | CYP1A2-substrate: | 0.084 |
| CYP2C19-inhibitor: | 0.037 | CYP2C19-substrate: | 0.714 |
| CYP2C9-inhibitor: | 0.026 | CYP2C9-substrate: | 0.087 |
| CYP2D6-inhibitor: | 0.012 | CYP2D6-substrate: | 0.128 |
| CYP3A4-inhibitor: | 0.045 | CYP3A4-substrate: | 0.268 |
——————————————————————————————————————————
ADMET: Excretion
| Clearance (CL): | 4.907 | Half-life (T1/2): | 0.728 |
——————————————————————————————————————————
ADMET: Toxicity
| hERG Blockers: | 0.005 | Human Hepatotoxicity (H-HT): | 0.308 |
| Drug-inuced Liver Injury (DILI): | 0.24 | AMES Toxicity: | 0.04 |
| Rat Oral Acute Toxicity: | 0.155 | Maximum Recommended Daily Dose: | 0.017 |
| Skin Sensitization: | 0.039 | Carcinogencity: | 0.035 |
| Eye Corrosion: | 0.004 | Eye Irritation: | 0.018 |
| Respiratory Toxicity: | 0.092 |