NPs Basic Information

|
Name |
Rosellichalasin
|
| Molecular Formula | C28H33NO5 | |
| IUPAC Name* |
(5Z,10E)-18-benzyl-6,8,15,16-tetramethyl-2,14-dioxa-19-azatetracyclo[10.8.0.01,17.013,15]icosa-5,10-diene-3,7,20-trione
|
|
| SMILES |
CC1C/C=C/C2C3C(O3)(C(C4C2(C(=O)NC4CC5=CC=CC=C5)OC(=O)C/C=C(\C1=O)/C)C)C
|
|
| InChI |
InChI=1S/C28H33NO5/c1-16-9-8-12-20-25-27(4,34-25)18(3)23-21(15-19-10-6-5-7-11-19)29-26(32)28(20,23)33-22(30)14-13-17(2)24(16)31/h5-8,10-13,16,18,20-21,23,25H,9,14-15H2,1-4H3,(H,29,32)/b12-8+,17-13-
|
|
| InChIKey |
XNIXILZHFOBLTQ-QCNUPDNZSA-N
|
|
| Synonyms |
Rosellichalasin; 123452-64-2; (5Z,10E)-18-benzyl-6,8,15,16-tetramethyl-2,14-dioxa-19-azatetracyclo[10.8.0.01,17.013,15]icosa-5,10-diene-3,7,20-trione; 3H-Oxacyclododecino(2,3-d)oxireno(f)isoindole-5,9,11(4H,8H,12H)-trione, 13,13a,14,14a,15a,15b-hexahydro-4,6,14,14a-tetramethyl-13-(phenylmethyl)-, (4S-(1E,4R*,6E,10aR*,13R*,13aR*,14R*,14aS*,15aR*,15bR*))-
|
|
| CAS | 123452-64-2 | |
| PubChem CID | 6442449 | |
| ChEMBL ID | NA |
*Note: the IUPAC Name was collected from PubChem.
Chemical Classification: |
|
|
|---|
——————————————————————————————————————————
NPs Species Source
| Endophyte ID | Endophyte Name | Family | Genus | Taxonomy ID | GenBank ID | Closest GenBank ID | Reference | |
|---|---|---|---|---|---|---|---|---|
| Endophyte ID | Endophyte Name | Family | Genus | Taxonomy ID | GenBank ID | Closest GenBank ID | Reference |
NPs Biological Activity
| Bioactivity Name | Target ID | Target Name | Target Type | Target Organism | Target Organism ID | Potency of Bioactivity | Activity Type | Value | Unit | Endophyte ID | Endophyte Name | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Bioactivity Name | Target ID | Target Name | Target Type | Target Organism | Target Organism ID | Potency of Bioactivity | Activity Type | Value | Unit | Endophyte ID | Endophyte Name |
NPs Physi-Chem Properties
| Molecular Weight: | 463.6 | ALogp: | 4.0 |
| HBD: | 1 | HBA: | 5 |
| Rotatable Bonds: | 2 | Lipinski's rule of five: | Accepted |
| Polar Surface Area: | 85.0 | Aromatic Rings: | 5 |
| Heavy Atoms: | 34 | QED Weighted: | 0.402 |
——————————————————————————————————————————
NPs ADMET Properties*
ADMET: Absorption
| Caco-2 Permeability: | -4.756 | MDCK Permeability: | 0.00006110 |
| Pgp-inhibitor: | 0.998 | Pgp-substrate: | 0.002 |
| Human Intestinal Absorption (HIA): | 0.171 | 20% Bioavailability (F20%): | 0.026 |
| 30% Bioavailability (F30%): | 0.392 |
——————————————————————————————————————————
ADMET: Distribution
| Blood-Brain-Barrier Penetration (BBB): | 0.441 | Plasma Protein Binding (PPB): | 96.20% |
| Volume Distribution (VD): | 1.512 | Fu: | 2.52% |
——————————————————————————————————————————
ADMET: Metabolism
| CYP1A2-inhibitor: | 0.024 | CYP1A2-substrate: | 0.237 |
| CYP2C19-inhibitor: | 0.543 | CYP2C19-substrate: | 0.332 |
| CYP2C9-inhibitor: | 0.387 | CYP2C9-substrate: | 0.083 |
| CYP2D6-inhibitor: | 0.076 | CYP2D6-substrate: | 0.337 |
| CYP3A4-inhibitor: | 0.929 | CYP3A4-substrate: | 0.668 |
——————————————————————————————————————————
ADMET: Excretion
| Clearance (CL): | 6.892 | Half-life (T1/2): | 0.081 |
——————————————————————————————————————————
ADMET: Toxicity
| hERG Blockers: | 0.326 | Human Hepatotoxicity (H-HT): | 0.825 |
| Drug-inuced Liver Injury (DILI): | 0.066 | AMES Toxicity: | 0.038 |
| Rat Oral Acute Toxicity: | 0.947 | Maximum Recommended Daily Dose: | 0.915 |
| Skin Sensitization: | 0.132 | Carcinogencity: | 0.063 |
| Eye Corrosion: | 0.003 | Eye Irritation: | 0.007 |
| Respiratory Toxicity: | 0.877 |
——————————————————————————————————————————
*Note: the ADMET properties was calculated by ADMETlab 2.0. Reference: PMID: 33893803.
Similar Compounds*
Compounds similar to EMNPD with top10 similarity:
| Similar NPs | Similar Drugs | ||||||
|---|---|---|---|---|---|---|---|
| NPs ID | NPs 2D Structure | Similarity Score | TTD ID | Drug 2D Structure | Similarity Score | ||
| ENC001756 |  |
0.696 | D0V3ZA | 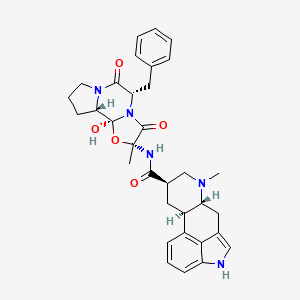 |
0.267 | ||
| ENC004801 |  |
0.688 | D01TSI | 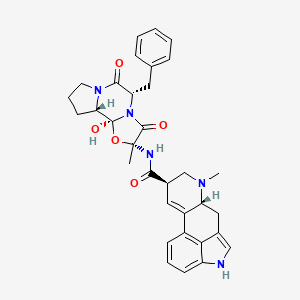 |
0.260 | ||
| ENC002419 |  |
0.630 | D09NNH | 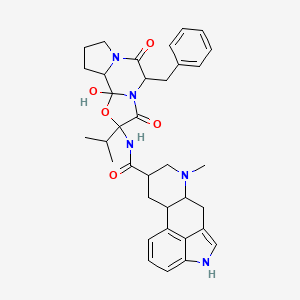 |
0.260 | ||
| ENC003802 |  |
0.593 | D0SP3D |  |
0.260 | ||
| ENC005546 |  |
0.574 | D08UMH | 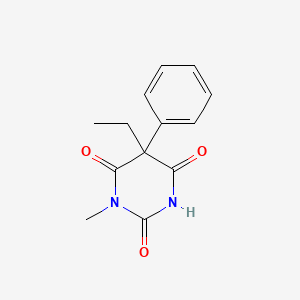 |
0.252 | ||
| ENC002828 |  |
0.573 | D0Y7RW | 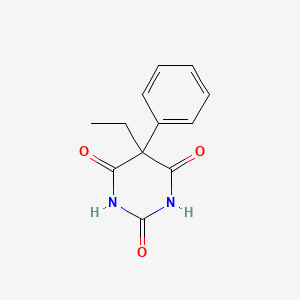 |
0.246 | ||
| ENC002202 |  |
0.563 | D0W7RJ | 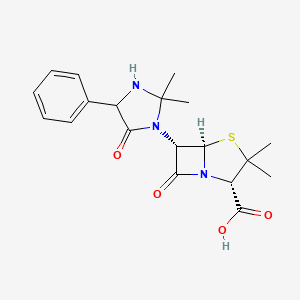 |
0.244 | ||
| ENC003229 |  |
0.562 | D0IN7I | 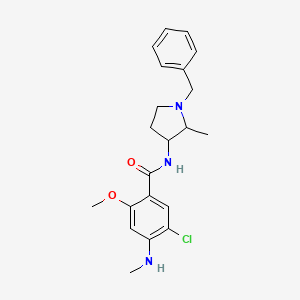 |
0.239 | ||
| ENC003712 |  |
0.559 | D0Z9NZ |  |
0.239 | ||
| ENC003672 |  |
0.553 | D06CWH | 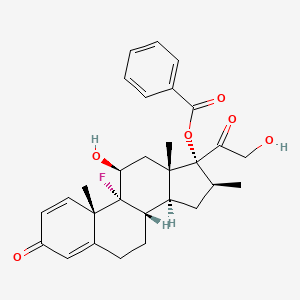 |
0.236 | ||