NPs Basic Information

|
Name |
Caryophyllene oxide
|
| Molecular Formula | C15H24O | |
| IUPAC Name* |
(1R,4R,6R,10S)-4,12,12-trimethyl-9-methylidene-5-oxatricyclo[8.2.0.04,6]dodecane
|
|
| SMILES |
C[C@@]12CC[C@@H]3[C@H](CC3(C)C)C(=C)CC[C@H]1O2
|
|
| InChI |
InChI=1S/C15H24O/c1-10-5-6-13-15(4,16-13)8-7-12-11(10)9-14(12,2)3/h11-13H,1,5-9H2,2-4H3/t11-,12-,13-,15-/m1/s1
|
|
| InChIKey |
NVEQFIOZRFFVFW-RGCMKSIDSA-N
|
|
| Synonyms |
Caryophyllene oxide; (-)-Caryophyllene oxide; 1139-30-6; beta-Caryophyllene oxide; beta-Caryophyllene epoxide; (-)-Epoxycaryophyllene; (-)Carophyllene oxide; trans-caryophyllene oxide; Epoxycaryophyllene; CHEMBL508894; S2XU9K448U; CHEBI:67818; .beta.-Caryophyllene oxide; (1R,4R,6R,10S)-4,12,12-trimethyl-9-methylidene-5-oxatricyclo[8.2.0.04,6]dodecane; (-)-Epoxydihydrocaryophyllene; Caryophyllene Oxide 1000 microg/mL in Isopropanol; (-)-.beta.-Caryophyllene epoxide; (-)-BETA-CARYOPHYLLENE EPOXIDE; (1R,4R,6R,10S)-4,12,12-TRIMETHYL-9-METHYLIDENE-5-OXATRICYCLO[8.2.0.0?,?]DODECANE; 5-Oxatricyclo(8.2.0.0(4,6))dodecane, 4,12,12-trimethyl-9-methylene-, (1R,4R,6R,10S)-; 5-Oxatricyclo(8.2.0.04,6)dodecane, 4,12,12-trimethyl-9-methylene-, (1R,4R,6R,10S)-; MFCD00134216; UNII-S2XU9K448U; HSDB 5466; EINECS 214-519-7; trimethyl(methylene)[?]; DSSTox_CID_30138; DSSTox_GSID_51586; CARYOPHYLLENE, EPOXIDE; SCHEMBL127077; DTXSID4051586; FEMA NO. 4085; (-)-Caryophyllene oxide, 95%; HY-N3544; ZINC2083320; Tox21_303807; 4,11,11-Trimethyl-8-methylene-5-oxatricyclo(8.2.0.0(4,6))dodecane; BDBM50241720; s3983; AKOS030241571; CCG-208462; 4,12,12-Trimethyl-9-methylene-5-oxatricyclo(8.2.0.04,6)dodecane, (1R,4R,6R,10S)-; NCGC00357089-01; AS-58060; .BETA.-CARYOPHYLLENE OXIDE [FHFI]; CAS-1139-30-6; 4.BETA.,5.ALPHA.-EPOXYCARYOPHYLLENE; CS-0023810; CARYOPHYLLENE 4.BETA.,5.ALPHA.-OXIDE; (-)-BETA-CARYOPHYLLENE EPOXIDE [HSDB]; C16908; CARYOPHYLLENE 4.BETA.,5.ALPHA.-EPOXIDE; E80731; SR-05000002236; SR-05000002236-2; W-108611; (-)-Caryophyllene oxide, analytical reference material; (1R,4R,5R,9S)-4,5-epoxycaryophyllan-8(13)-ene; Q27136294; (-)-Caryophyllene oxide, >=99.0% (sum of enantiomers, GC); (1R,4R,6R,10S)-9-Methylene-4,12,12-trimethyl-5-oxatricyclo[8.2.0.0(4,6])dodecane; 4-12,12-TRIMETHYL-9-METHYLENE-5-OXATRICYLO (8.2.0.04,6) DODECANE; (1R-(1R*,4R*,6R*,10S*))-4,12,12-Trimethyl-9-methylene-5-oxatricyclo(8.2.0.04,6)dodecane; 105120-46-5; 5-Oxatricyclo(8.2.0.0(sup 4,6))dodecane, 4,12,12-trimethyl-9-methylene-, (1R,4R,6R,10S)-; 5-OXATRICYCLO(8.2.0.04,6)DODECANE, 4,12,12-TRIMETHYL-9-METHYLENE-, (1R-(1R*,4R*,6R*,10S*))-
|
|
| CAS | 1139-30-6 | |
| PubChem CID | 1742210 | |
| ChEMBL ID | CHEMBL508894 |
*Note: the IUPAC Name was collected from PubChem.
Chemical Classification: |
|
|
|---|
——————————————————————————————————————————
NPs Species Source
| Endophyte ID | Endophyte Name | Family | Genus | Taxonomy ID | GenBank ID | Closest GenBank ID | Reference | |
|---|---|---|---|---|---|---|---|---|
| Endophyte ID | Endophyte Name | Family | Genus | Taxonomy ID | GenBank ID | Closest GenBank ID | Reference |
NPs Biological Activity
| Bioactivity Name | Target ID | Target Name | Target Type | Target Organism | Target Organism ID | Potency of Bioactivity | Activity Type | Value | Unit | Endophyte ID | Endophyte Name | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Bioactivity Name | Target ID | Target Name | Target Type | Target Organism | Target Organism ID | Potency of Bioactivity | Activity Type | Value | Unit | Endophyte ID | Endophyte Name |
NPs Physi-Chem Properties
| Molecular Weight: | 220.35 | ALogp: | 3.6 |
| HBD: | 0 | HBA: | 1 |
| Rotatable Bonds: | 0 | Lipinski's rule of five: | Accepted |
| Polar Surface Area: | 12.5 | Aromatic Rings: | 3 |
| Heavy Atoms: | 16 | QED Weighted: | 0.43 |
——————————————————————————————————————————
NPs ADMET Properties*
ADMET: Absorption
| Caco-2 Permeability: | -4.673 | MDCK Permeability: | 0.00002050 |
| Pgp-inhibitor: | 0.001 | Pgp-substrate: | 0 |
| Human Intestinal Absorption (HIA): | 0.003 | 20% Bioavailability (F20%): | 0.338 |
| 30% Bioavailability (F30%): | 0.01 |
——————————————————————————————————————————
ADMET: Distribution
| Blood-Brain-Barrier Penetration (BBB): | 0.249 | Plasma Protein Binding (PPB): | 82.27% |
| Volume Distribution (VD): | 1.279 | Fu: | 18.29% |
——————————————————————————————————————————
ADMET: Metabolism
| CYP1A2-inhibitor: | 0.172 | CYP1A2-substrate: | 0.549 |
| CYP2C19-inhibitor: | 0.177 | CYP2C19-substrate: | 0.916 |
| CYP2C9-inhibitor: | 0.267 | CYP2C9-substrate: | 0.383 |
| CYP2D6-inhibitor: | 0.016 | CYP2D6-substrate: | 0.809 |
| CYP3A4-inhibitor: | 0.077 | CYP3A4-substrate: | 0.339 |
——————————————————————————————————————————
ADMET: Excretion
| Clearance (CL): | 6.458 | Half-life (T1/2): | 0.17 |
——————————————————————————————————————————
ADMET: Toxicity
| hERG Blockers: | 0.046 | Human Hepatotoxicity (H-HT): | 0.281 |
| Drug-inuced Liver Injury (DILI): | 0.397 | AMES Toxicity: | 0.014 |
| Rat Oral Acute Toxicity: | 0.071 | Maximum Recommended Daily Dose: | 0.816 |
| Skin Sensitization: | 0.413 | Carcinogencity: | 0.149 |
| Eye Corrosion: | 0.597 | Eye Irritation: | 0.774 |
| Respiratory Toxicity: | 0.873 |
——————————————————————————————————————————
*Note: the ADMET properties was calculated by ADMETlab 2.0. Reference: PMID: 33893803.
Similar Compounds*
Compounds similar to EMNPD with top10 similarity:
| Similar NPs | Similar Drugs | ||||||
|---|---|---|---|---|---|---|---|
| NPs ID | NPs 2D Structure | Similarity Score | TTD ID | Drug 2D Structure | Similarity Score | ||
| ENC001297 |  |
0.527 | D0C7JF | 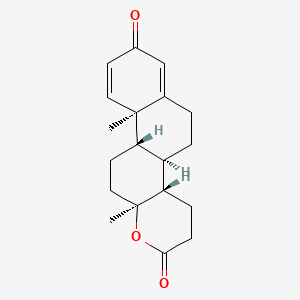 |
0.293 | ||
| ENC001630 |  |
0.491 | D0L2LS | 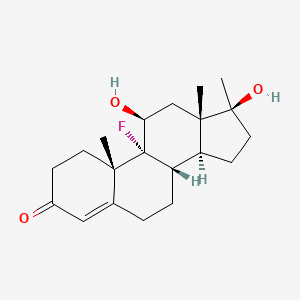 |
0.282 | ||
| ENC001565 |  |
0.491 | D0D2VS |  |
0.280 | ||
| ENC001563 |  |
0.491 | D0U3GL | 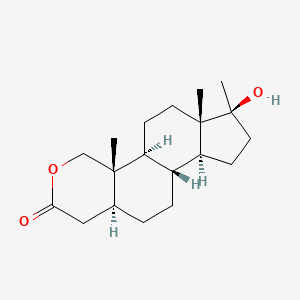 |
0.280 | ||
| ENC001826 |  |
0.491 | D0Z1XD | 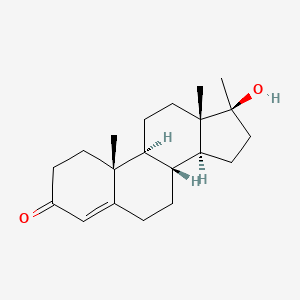 |
0.280 | ||
| ENC002256 |  |
0.450 | D04VIS |  |
0.273 | ||
| ENC001663 |  |
0.441 | D0S3WH |  |
0.266 | ||
| ENC002543 |  |
0.441 | D0H1QY |  |
0.259 | ||
| ENC002110 |  |
0.393 | D0K0EK |  |
0.256 | ||
| ENC003084 |  |
0.393 | D06XMU |  |
0.256 | ||