NPs Basic Information
|
Name |
Ginsenoside Rh2
|
| Molecular Formula | C36H62O8 | |
| IUPAC Name* |
(2R,3R,4S,5S,6R)-2-[[(3S,5R,8R,9R,10R,12R,13R,14R,17S)-12-hydroxy-17-[(2S)-2-hydroxy-6-methylhept-5-en-2-yl]-4,4,8,10,14-pentamethyl-2,3,5,6,7,9,11,12,13,15,16,17-dodecahydro-1H-cyclopenta[a]phenanthren-3-yl]oxy]-6-(hydroxymethyl)oxane-3,4,5-triol
|
|
| SMILES |
CC(=CCC[C@@](C)([C@H]1CC[C@@]2([C@@H]1[C@@H](C[C@H]3[C@]2(CC[C@@H]4[C@@]3(CC[C@@H](C4(C)C)O[C@H]5[C@@H]([C@H]([C@@H]([C@H](O5)CO)O)O)O)C)C)O)C)O)C
|
|
| InChI |
InChI=1S/C36H62O8/c1-20(2)10-9-14-36(8,42)21-11-16-35(7)27(21)22(38)18-25-33(5)15-13-26(32(3,4)24(33)12-17-34(25,35)6)44-31-30(41)29(40)28(39)23(19-37)43-31/h10,21-31,37-42H,9,11-19H2,1-8H3/t21-,22+,23+,24-,25+,26-,27-,28+,29-,30+,31-,33-,34+,35+,36-/m0/s1
|
|
| InChIKey |
CKUVNOCSBYYHIS-IRFFNABBSA-N
|
|
| Synonyms |
Ginsenoside Rh2; 78214-33-2; 20(S)-Ginsenoside Rh2; (20S)-ginsenoside Rh2; 20(S)-Ginsenoside; Ginsenoside-Rh2; CHEBI:77147; 20S-Ginsenoside Rh2; 0JU44A5KWG; (2R,3R,4S,5S,6R)-2-[[(3S,5R,8R,9R,10R,12R,13R,14R,17S)-12-hydroxy-17-[(2S)-2-hydroxy-6-methylhept-5-en-2-yl]-4,4,8,10,14-pentamethyl-2,3,5,6,7,9,11,12,13,15,16,17-dodecahydro-1H-cyclopenta[a]phenanthren-3-yl]oxy]-6-(hydroxymethyl)oxane-3,4,5-triol; (20R)-Ginsenoside Rh2; 20(S)-Rh2; (3beta,12beta)-12,20-dihydroxydammar-24-en-3-yl beta-D-glucopyranoside; (20R)Ginsenoside Rh2; GinsenosideRh2; 112246-15-8; (S)Ginsenoside-Rh2; 20S-GinsenosideRh2; UNII-0JU44A5KWG; GINSENOSIDE RH2(S); 20(S)- Ginsenoside Rh2; GINSENOSIDE 20-RH2; CHEMBL1783834; DTXSID70999457; 67400-17-3; HY-N0605; BDBM50023457; Ginsenoside Rh2, analytical standard; MFCD00800712; s9023; s9036; ZINC72129809; AKOS037514675; CCG-270259; CCG-270261; CS-3835; AC-33940; Ginsenoside Rh2; 20(s)-Ginsenoside Rh2; C22128; 20S-protopanaxdiol-3-O-beta-D- glucopyranoside; 214G332; Q-100827; Q27146703; 3-O-.BETA.-D-GLUCOPYRANOSYL-20(S)-PROTOPANAXADIOL; 3beta-(beta-D-glucopyranosyloxy)dammar-24-ene-3beta,20beta-diol; beta-D-Glucopyranoside, (3beta,12beta)-12,20-dihydroxydammar-24-en-3-yl; .BETA.-D-GLUCOPYRANOSIDE, (3.BETA.,12.BETA.)-12,20-DIHYDROXYDAMMAR-24-EN-3-YL
|
|
| CAS | 78214-33-2 | |
| PubChem CID | 119307 | |
| ChEMBL ID | CHEMBL1783834 |
*Note: the IUPAC Name was collected from PubChem.
Chemical Classification: |
|
|
|---|
——————————————————————————————————————————
NPs Species Source
| Endophyte ID | Endophyte Name | Family | Genus | Taxonomy ID | GenBank ID | Closest GenBank ID | Reference | |
|---|---|---|---|---|---|---|---|---|
| Endophyte ID | Endophyte Name | Family | Genus | Taxonomy ID | GenBank ID | Closest GenBank ID | Reference |
NPs Biological Activity
| Bioactivity Name | Target ID | Target Name | Target Type | Target Organism | Target Organism ID | Potency of Bioactivity | Activity Type | Value | Unit | Endophyte ID | Endophyte Name | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Bioactivity Name | Target ID | Target Name | Target Type | Target Organism | Target Organism ID | Potency of Bioactivity | Activity Type | Value | Unit | Endophyte ID | Endophyte Name |
NPs Physi-Chem Properties
| Molecular Weight: | 622.9 | ALogp: | 5.6 |
| HBD: | 6 | HBA: | 8 |
| Rotatable Bonds: | 7 | Lipinski's rule of five: | Rejected |
| Polar Surface Area: | 140.0 | Aromatic Rings: | 5 |
| Heavy Atoms: | 44 | QED Weighted: | 0.175 |
——————————————————————————————————————————
NPs ADMET Properties*
ADMET: Absorption
| Caco-2 Permeability: | -5.074 | MDCK Permeability: | 0.00001770 |
| Pgp-inhibitor: | 0.993 | Pgp-substrate: | 0.01 |
| Human Intestinal Absorption (HIA): | 0.03 | 20% Bioavailability (F20%): | 0.623 |
| 30% Bioavailability (F30%): | 0.932 |
——————————————————————————————————————————
ADMET: Distribution
| Blood-Brain-Barrier Penetration (BBB): | 0.004 | Plasma Protein Binding (PPB): | 88.49% |
| Volume Distribution (VD): | 0.792 | Fu: | 5.76% |
——————————————————————————————————————————
ADMET: Metabolism
| CYP1A2-inhibitor: | 0.004 | CYP1A2-substrate: | 0.13 |
| CYP2C19-inhibitor: | 0.007 | CYP2C19-substrate: | 0.815 |
| CYP2C9-inhibitor: | 0.043 | CYP2C9-substrate: | 0.058 |
| CYP2D6-inhibitor: | 0.002 | CYP2D6-substrate: | 0.086 |
| CYP3A4-inhibitor: | 0.121 | CYP3A4-substrate: | 0.152 |
——————————————————————————————————————————
ADMET: Excretion
| Clearance (CL): | 1.439 | Half-life (T1/2): | 0.352 |
——————————————————————————————————————————
ADMET: Toxicity
| hERG Blockers: | 0.083 | Human Hepatotoxicity (H-HT): | 0.213 |
| Drug-inuced Liver Injury (DILI): | 0.009 | AMES Toxicity: | 0.015 |
| Rat Oral Acute Toxicity: | 0.052 | Maximum Recommended Daily Dose: | 0.066 |
| Skin Sensitization: | 0.759 | Carcinogencity: | 0.009 |
| Eye Corrosion: | 0.004 | Eye Irritation: | 0.051 |
| Respiratory Toxicity: | 0.916 |
——————————————————————————————————————————
*Note: the ADMET properties was calculated by ADMETlab 2.0. Reference: PMID: 33893803.
Similar Compounds*
Compounds similar to EMNPD with top10 similarity:
| Similar NPs | Similar Drugs | ||||||
|---|---|---|---|---|---|---|---|
| NPs ID | NPs 2D Structure | Similarity Score | TTD ID | Drug 2D Structure | Similarity Score | ||
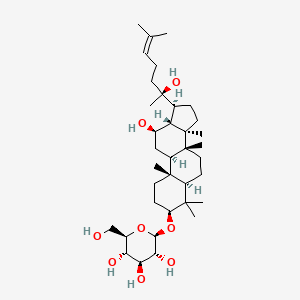
| ENC001939 | 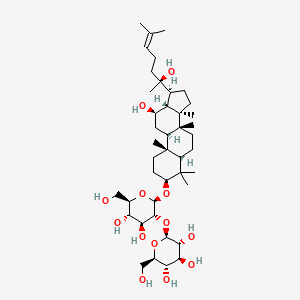 |
0.797 | D03MTN | 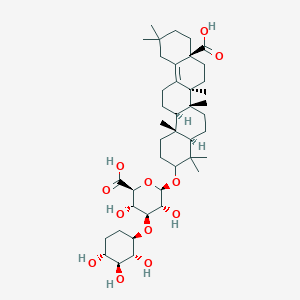 |
0.404 | ||
| ENC002246 | 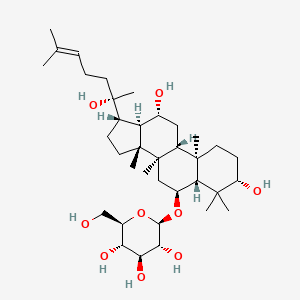 |
0.776 | D04RYU | 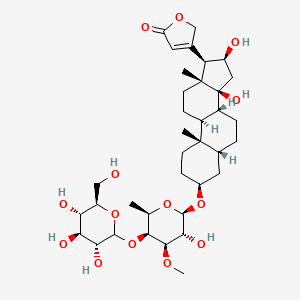 |
0.354 | ||
| ENC001938 | 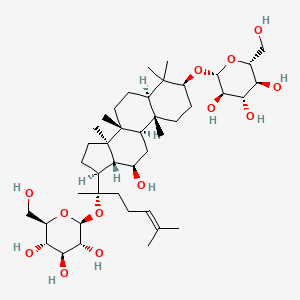 |
0.739 | D07QQD | 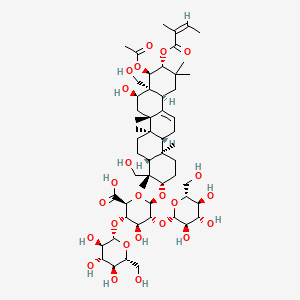 |
0.347 | ||
| ENC001918 | 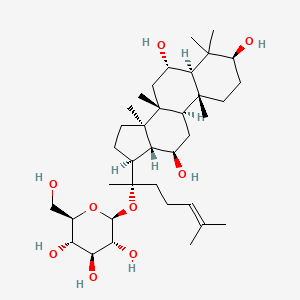 |
0.688 | D0P2IT | 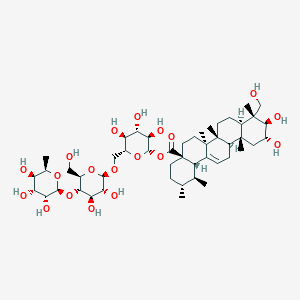 |
0.341 | ||
| ENC002655 | 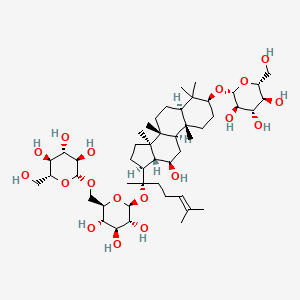 |
0.617 | D04MRG | 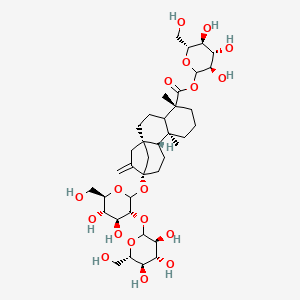 |
0.335 | ||
| ENC002180 | 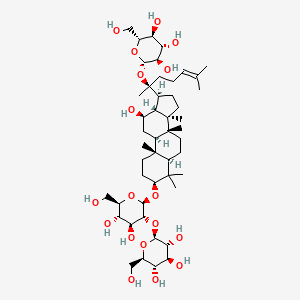 |
0.617 | D0X7XG |  |
0.331 | ||
| ENC002152 |  |
0.600 | D0AR3J |  |
0.322 | ||
| ENC002245 |  |
0.543 | D0S0NK |  |
0.309 | ||
| ENC001894 |  |
0.543 | D09HTS |  |
0.306 | ||
| ENC001933 |  |
0.531 | D07ORO |  |
0.299 | ||