NPs Basic Information

|
Name |
(-)-acrozine C
|
| Molecular Formula | C16H19N3O4 | |
| IUPAC Name* |
(3R,6R)-3-(1H-indol-3-ylmethyl)-1,3-dimethoxy-6-methylpiperazine-2,5-dione
|
|
| SMILES |
C[C@@H]1C(=O)N[C@](C(=O)N1OC)(CC2=CNC3=CC=CC=C32)OC
|
|
| InChI |
InChI=1S/C16H19N3O4/c1-10-14(20)18-16(22-2,15(21)19(10)23-3)8-11-9-17-13-7-5-4-6-12(11)13/h4-7,9-10,17H,8H2,1-3H3,(H,18,20)/t10-,16-/m1/s1
|
|
| InChIKey |
OZHUNWPUOSMOEE-QLJPJBMISA-N
|
|
| Synonyms |
(-)-acrozine C
|
|
| CAS | NA | |
| PubChem CID | 156582033 | |
| ChEMBL ID | NA |
*Note: the IUPAC Name was collected from PubChem.
Chemical Classification: |
|
|
|---|
——————————————————————————————————————————
NPs Species Source
| Endophyte ID | Endophyte Name | Family | Genus | Taxonomy ID | GenBank ID | Closest GenBank ID | Reference | |
|---|---|---|---|---|---|---|---|---|
| Endophyte ID | Endophyte Name | Family | Genus | Taxonomy ID | GenBank ID | Closest GenBank ID | Reference |
NPs Biological Activity
| Bioactivity Name | Target ID | Target Name | Target Type | Target Organism | Target Organism ID | Potency of Bioactivity | Activity Type | Value | Unit | Endophyte ID | Endophyte Name | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Bioactivity Name | Target ID | Target Name | Target Type | Target Organism | Target Organism ID | Potency of Bioactivity | Activity Type | Value | Unit | Endophyte ID | Endophyte Name |
NPs Physi-Chem Properties
| Molecular Weight: | 317.34 | ALogp: | 1.2 |
| HBD: | 2 | HBA: | 4 |
| Rotatable Bonds: | 4 | Lipinski's rule of five: | Accepted |
| Polar Surface Area: | 83.7 | Aromatic Rings: | 3 |
| Heavy Atoms: | 23 | QED Weighted: | 0.892 |
——————————————————————————————————————————
NPs ADMET Properties*
ADMET: Absorption
| Caco-2 Permeability: | -4.496 | MDCK Permeability: | 0.00001260 |
| Pgp-inhibitor: | 0.459 | Pgp-substrate: | 0.41 |
| Human Intestinal Absorption (HIA): | 0.009 | 20% Bioavailability (F20%): | 0.008 |
| 30% Bioavailability (F30%): | 0.911 |
——————————————————————————————————————————
ADMET: Distribution
| Blood-Brain-Barrier Penetration (BBB): | 0.787 | Plasma Protein Binding (PPB): | 71.84% |
| Volume Distribution (VD): | 0.691 | Fu: | 39.98% |
——————————————————————————————————————————
ADMET: Metabolism
| CYP1A2-inhibitor: | 0.035 | CYP1A2-substrate: | 0.925 |
| CYP2C19-inhibitor: | 0.359 | CYP2C19-substrate: | 0.798 |
| CYP2C9-inhibitor: | 0.191 | CYP2C9-substrate: | 0.328 |
| CYP2D6-inhibitor: | 0.01 | CYP2D6-substrate: | 0.127 |
| CYP3A4-inhibitor: | 0.183 | CYP3A4-substrate: | 0.928 |
——————————————————————————————————————————
ADMET: Excretion
| Clearance (CL): | 7.947 | Half-life (T1/2): | 0.842 |
——————————————————————————————————————————
ADMET: Toxicity
| hERG Blockers: | 0.008 | Human Hepatotoxicity (H-HT): | 0.349 |
| Drug-inuced Liver Injury (DILI): | 0.958 | AMES Toxicity: | 0.038 |
| Rat Oral Acute Toxicity: | 0.418 | Maximum Recommended Daily Dose: | 0.485 |
| Skin Sensitization: | 0.192 | Carcinogencity: | 0.039 |
| Eye Corrosion: | 0.003 | Eye Irritation: | 0.01 |
| Respiratory Toxicity: | 0.016 |
——————————————————————————————————————————
*Note: the ADMET properties was calculated by ADMETlab 2.0. Reference: PMID: 33893803.
Similar Compounds*
Compounds similar to EMNPD with top10 similarity:
| Similar NPs | Similar Drugs | ||||||
|---|---|---|---|---|---|---|---|
| NPs ID | NPs 2D Structure | Similarity Score | TTD ID | Drug 2D Structure | Similarity Score | ||
| ENC004346 |  |
1.000 | D05EJG | 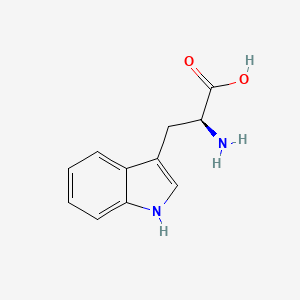 |
0.359 | ||
| ENC005918 |  |
1.000 | D07RGW | 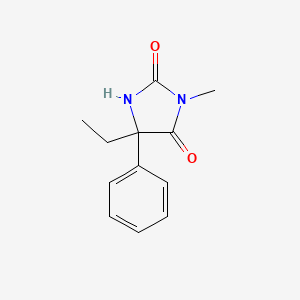 |
0.317 | ||
| ENC004343 |  |
0.598 | D0E3WQ |  |
0.309 | ||
| ENC004345 |  |
0.598 | D08UMH | 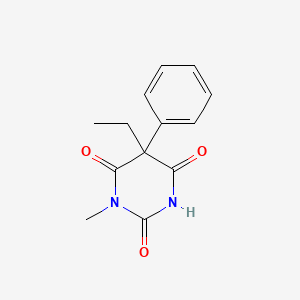 |
0.299 | ||
| ENC004342 |  |
0.598 | D0K0KH |  |
0.289 | ||
| ENC004344 |  |
0.598 | D00YLW |  |
0.288 | ||
| ENC005916 |  |
0.598 | D0AN7B | 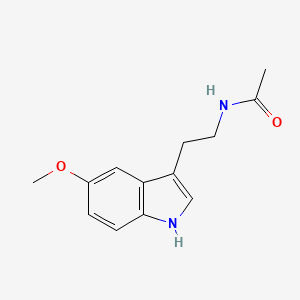 |
0.287 | ||
| ENC005917 |  |
0.598 | D08EOD | 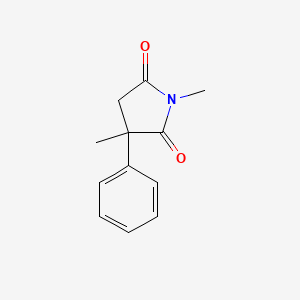 |
0.280 | ||
| ENC003991 |  |
0.488 | D02DMQ | 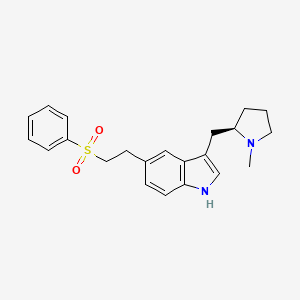 |
0.279 | ||
| ENC004869 |  |
0.477 | D0W7WC |  |
0.279 | ||