NPs Basic Information

|
Name |
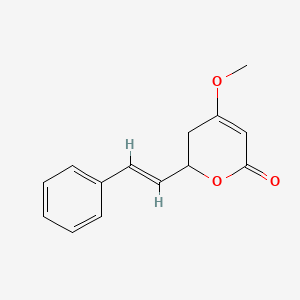
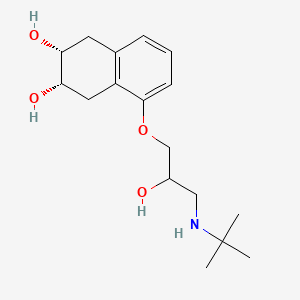
Photipyrone B
|
| Molecular Formula | C11H18O5 | |
| IUPAC Name* |
(2S)-2-[(1S)-1,3-dihydroxypentyl]-4-methoxy-2,3-dihydropyran-6-one
|
|
| SMILES |
CCC(C[C@@H]([C@@H]1CC(=CC(=O)O1)OC)O)O
|
|
| InChI |
InChI=1S/C11H18O5/c1-3-7(12)4-9(13)10-5-8(15-2)6-11(14)16-10/h6-7,9-10,12-13H,3-5H2,1-2H3/t7?,9-,10-/m0/s1
|
|
| InChIKey |
QPHDBTAGXJAYBS-IVNRZZHDSA-N
|
|
| Synonyms |
Photipyrone B
|
|
| CAS | NA | |
| PubChem CID | 59052169 | |
| ChEMBL ID | NA |
*Note: the IUPAC Name was collected from PubChem.
Chemical Classification: |
|
|
|---|
——————————————————————————————————————————
NPs Physi-Chem Properties
| Molecular Weight: | 230.26 | ALogp: | 0.5 |
| HBD: | 2 | HBA: | 5 |
| Rotatable Bonds: | 5 | Lipinski's rule of five: | Accepted |
| Polar Surface Area: | 76.0 | Aromatic Rings: | 1 |
| Heavy Atoms: | 16 | QED Weighted: | 0.682 |
——————————————————————————————————————————
NPs ADMET Properties*
ADMET: Absorption
| Caco-2 Permeability: | -4.722 | MDCK Permeability: | 0.00015685 |
| Pgp-inhibitor: | 0.005 | Pgp-substrate: | 0.55 |
| Human Intestinal Absorption (HIA): | 0.483 | 20% Bioavailability (F20%): | 0.021 |
| 30% Bioavailability (F30%): | 0.939 |
——————————————————————————————————————————
ADMET: Distribution
| Blood-Brain-Barrier Penetration (BBB): | 0.435 | Plasma Protein Binding (PPB): | 16.91% |
| Volume Distribution (VD): | 0.639 | Fu: | 67.95% |
——————————————————————————————————————————
ADMET: Metabolism
| CYP1A2-inhibitor: | 0.021 | CYP1A2-substrate: | 0.12 |
| CYP2C19-inhibitor: | 0.019 | CYP2C19-substrate: | 0.807 |
| CYP2C9-inhibitor: | 0.007 | CYP2C9-substrate: | 0.094 |
| CYP2D6-inhibitor: | 0.004 | CYP2D6-substrate: | 0.197 |
| CYP3A4-inhibitor: | 0.016 | CYP3A4-substrate: | 0.389 |
——————————————————————————————————————————
ADMET: Excretion
| Clearance (CL): | 10.212 | Half-life (T1/2): | 0.889 |
——————————————————————————————————————————
ADMET: Toxicity
| hERG Blockers: | 0.026 | Human Hepatotoxicity (H-HT): | 0.569 |
| Drug-inuced Liver Injury (DILI): | 0.066 | AMES Toxicity: | 0.038 |
| Rat Oral Acute Toxicity: | 0.097 | Maximum Recommended Daily Dose: | 0.943 |
| Skin Sensitization: | 0.959 | Carcinogencity: | 0.51 |
| Eye Corrosion: | 0.394 | Eye Irritation: | 0.846 |
| Respiratory Toxicity: | 0.706 |