NPs Basic Information

|
Name |
Arugosin C
|
| Molecular Formula | C25H28O6 | |
| IUPAC Name* |
7,11-dihydroxy-17-(2-hydroxypropan-2-yl)-13-methyl-6-(3-methylbut-2-enyl)-2,15-dioxatetracyclo[8.7.1.03,8.014,18]octadeca-3(8),4,6,10,12,14(18)-hexaen-9-one
|
|
| SMILES |
CC1=CC(=C2C3=C1OCC(C3OC4=C(C2=O)C(=C(C=C4)CC=C(C)C)O)C(C)(C)O)O
|
|
| InChI |
InChI=1S/C25H28O6/c1-12(2)6-7-14-8-9-17-19(21(14)27)22(28)18-16(26)10-13(3)23-20(18)24(31-17)15(11-30-23)25(4,5)29/h6,8-10,15,24,26-27,29H,7,11H2,1-5H3
|
|
| InChIKey |
MYJGUMZTENHAAQ-UHFFFAOYSA-N
|
|
| Synonyms |
Arugosin C; 50875-10-0; CHEBI:68860; FD39J64RWK; UNII-FD39J64RWK; 6,8-dihydroxy-1-(2-hydroxypropan-2-yl)-4-methyl-9-(3-methylbut-2-en-1-yl)-1,12a-dihydrochromeno[4,5-bc][1]benzoxepin-7(2h)-one; MLS004257379; CHEMBL3092848; SMR003082511; Q15410264; 1,12a-Dihydro-6,8-dihydroxy-1-(1-hydroxy-1-methylethyl)-4-methyl-9-(3-methylbut-2-enyl)(1)benzopy rano(4,5-bc)(1)benzoxepin-7(2H)-one; 1,12a-Dihydro-6,8-dihydroxy-1-(1-hydroxy-1-methylethyl)-4-methyl-9-(3-methylbut-2-enyl)(1)benzopyrano(4,5-bc)(1)benzoxepin-7(2H)-one
|
|
| CAS | 50875-10-0 | |
| PubChem CID | 191158 | |
| ChEMBL ID | CHEMBL3092848 |
*Note: the IUPAC Name was collected from PubChem.
Chemical Classification: |
|
|
|---|
——————————————————————————————————————————
NPs Species Source
| Endophyte ID | Endophyte Name | Family | Genus | Taxonomy ID | GenBank ID | Closest GenBank ID | Reference | |
|---|---|---|---|---|---|---|---|---|
| Endophyte ID | Endophyte Name | Family | Genus | Taxonomy ID | GenBank ID | Closest GenBank ID | Reference |
NPs Biological Activity
| Bioactivity Name | Target ID | Target Name | Target Type | Target Organism | Target Organism ID | Potency of Bioactivity | Activity Type | Value | Unit | Endophyte ID | Endophyte Name | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Bioactivity Name | Target ID | Target Name | Target Type | Target Organism | Target Organism ID | Potency of Bioactivity | Activity Type | Value | Unit | Endophyte ID | Endophyte Name |
NPs Physi-Chem Properties
| Molecular Weight: | 424.5 | ALogp: | 5.0 |
| HBD: | 3 | HBA: | 6 |
| Rotatable Bonds: | 3 | Lipinski's rule of five: | Accepted |
| Polar Surface Area: | 96.2 | Aromatic Rings: | 4 |
| Heavy Atoms: | 31 | QED Weighted: | 0.607 |
——————————————————————————————————————————
NPs ADMET Properties*
ADMET: Absorption
| Caco-2 Permeability: | -4.983 | MDCK Permeability: | 0.00001390 |
| Pgp-inhibitor: | 0.145 | Pgp-substrate: | 0.005 |
| Human Intestinal Absorption (HIA): | 0.07 | 20% Bioavailability (F20%): | 0.132 |
| 30% Bioavailability (F30%): | 0.007 |
——————————————————————————————————————————
ADMET: Distribution
| Blood-Brain-Barrier Penetration (BBB): | 0.014 | Plasma Protein Binding (PPB): | 99.31% |
| Volume Distribution (VD): | 0.527 | Fu: | 1.98% |
——————————————————————————————————————————
ADMET: Metabolism
| CYP1A2-inhibitor: | 0.336 | CYP1A2-substrate: | 0.484 |
| CYP2C19-inhibitor: | 0.613 | CYP2C19-substrate: | 0.133 |
| CYP2C9-inhibitor: | 0.751 | CYP2C9-substrate: | 0.849 |
| CYP2D6-inhibitor: | 0.641 | CYP2D6-substrate: | 0.256 |
| CYP3A4-inhibitor: | 0.314 | CYP3A4-substrate: | 0.296 |
——————————————————————————————————————————
ADMET: Excretion
| Clearance (CL): | 3.786 | Half-life (T1/2): | 0.045 |
——————————————————————————————————————————
ADMET: Toxicity
| hERG Blockers: | 0.026 | Human Hepatotoxicity (H-HT): | 0.254 |
| Drug-inuced Liver Injury (DILI): | 0.894 | AMES Toxicity: | 0.343 |
| Rat Oral Acute Toxicity: | 0.567 | Maximum Recommended Daily Dose: | 0.473 |
| Skin Sensitization: | 0.449 | Carcinogencity: | 0.596 |
| Eye Corrosion: | 0.003 | Eye Irritation: | 0.608 |
| Respiratory Toxicity: | 0.36 |
——————————————————————————————————————————
*Note: the ADMET properties was calculated by ADMETlab 2.0. Reference: PMID: 33893803.
Similar Compounds*
Compounds similar to EMNPD with top10 similarity:
| Similar NPs | Similar Drugs | ||||||
|---|---|---|---|---|---|---|---|
| NPs ID | NPs 2D Structure | Similarity Score | TTD ID | Drug 2D Structure | Similarity Score | ||
| ENC004126 |  |
0.565 | D0Q0PR | 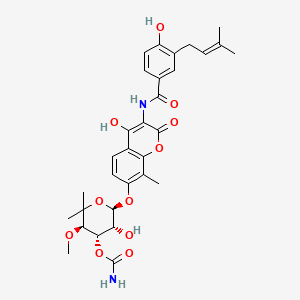 |
0.301 | ||
| ENC003568 |  |
0.509 | D07MGA |  |
0.261 | ||
| ENC003569 |  |
0.509 | D0AZ8C | 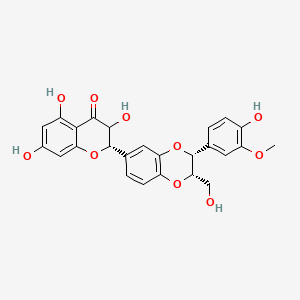 |
0.255 | ||
| ENC003968 |  |
0.509 | D04TDQ | 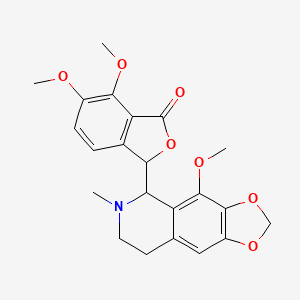 |
0.252 | ||
| ENC006093 |  |
0.482 | D0F7CS | 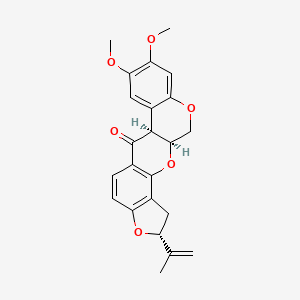 |
0.248 | ||
| ENC002916 |  |
0.482 | D01XWG |  |
0.247 | ||
| ENC002341 |  |
0.469 | D01XDL |  |
0.238 | ||
| ENC003963 |  |
0.462 | D07VLY |  |
0.234 | ||
| ENC003962 |  |
0.462 | D0C9XJ |  |
0.234 | ||
| ENC003942 |  |
0.453 | D0W6DG |  |
0.224 | ||