NPs Basic Information
|
Name |
1-Docosene
|
| Molecular Formula | C22H44 | |
| IUPAC Name* |
docos-1-ene
|
|
| SMILES |
CCCCCCCCCCCCCCCCCCCCC=C
|
|
| InChI |
InChI=1S/C22H44/c1-3-5-7-9-11-13-15-17-19-21-22-20-18-16-14-12-10-8-6-4-2/h3H,1,4-22H2,2H3
|
|
| InChIKey |
SPURMHFLEKVAAS-UHFFFAOYSA-N
|
|
| Synonyms |
1-Docosene; 1599-67-3; docos-1-ene; Docosene; AAM97MY7YB; NSC-78486; Alkenes, C20-24 .alpha.-; UNII-AAM97MY7YB; EINECS 216-490-6; NSC 78486; AI3-36497; 1-Docosene; >99%; EC 216-490-6; DTXSID5029219; CHEBI:84220; BAA59967; NSC78486; LMFA11000312; MFCD00027085; ZINC59674948; AKOS028108465; AS-56237; DB-043441; CS-0213243; D1013; FT-0633694; D89761; Q27157591
|
|
| CAS | 1599-67-3 | |
| PubChem CID | 74138 | |
| ChEMBL ID | NA |
*Note: the IUPAC Name was collected from PubChem.
Chemical Classification: |
|
|
|---|
——————————————————————————————————————————
NPs Species Source
| Endophyte ID | Endophyte Name | Family | Genus | Taxonomy ID | GenBank ID | Closest GenBank ID | Reference | |
|---|---|---|---|---|---|---|---|---|
| Endophyte ID | Endophyte Name | Family | Genus | Taxonomy ID | GenBank ID | Closest GenBank ID | Reference |
NPs Biological Activity
| Bioactivity Name | Target ID | Target Name | Target Type | Target Organism | Target Organism ID | Potency of Bioactivity | Activity Type | Value | Unit | Endophyte ID | Endophyte Name | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Bioactivity Name | Target ID | Target Name | Target Type | Target Organism | Target Organism ID | Potency of Bioactivity | Activity Type | Value | Unit | Endophyte ID | Endophyte Name |
NPs Physi-Chem Properties
| Molecular Weight: | 308.6 | ALogp: | 12.2 |
| HBD: | 0 | HBA: | 0 |
| Rotatable Bonds: | 19 | Lipinski's rule of five: | Rejected |
| Polar Surface Area: | 0.0 | Aromatic Rings: | 0 |
| Heavy Atoms: | 22 | QED Weighted: | 0.151 |
——————————————————————————————————————————
NPs ADMET Properties*
ADMET: Absorption
| Caco-2 Permeability: | -5.033 | MDCK Permeability: | 0.00000710 |
| Pgp-inhibitor: | 0 | Pgp-substrate: | 0 |
| Human Intestinal Absorption (HIA): | 0.003 | 20% Bioavailability (F20%): | 0.115 |
| 30% Bioavailability (F30%): | 0.947 |
——————————————————————————————————————————
ADMET: Distribution
| Blood-Brain-Barrier Penetration (BBB): | 0.033 | Plasma Protein Binding (PPB): | 100.71% |
| Volume Distribution (VD): | 3.915 | Fu: | 0.87% |
——————————————————————————————————————————
ADMET: Metabolism
| CYP1A2-inhibitor: | 0.1 | CYP1A2-substrate: | 0.162 |
| CYP2C19-inhibitor: | 0.237 | CYP2C19-substrate: | 0.057 |
| CYP2C9-inhibitor: | 0.058 | CYP2C9-substrate: | 0.959 |
| CYP2D6-inhibitor: | 0.257 | CYP2D6-substrate: | 0.112 |
| CYP3A4-inhibitor: | 0.287 | CYP3A4-substrate: | 0.034 |
——————————————————————————————————————————
ADMET: Excretion
| Clearance (CL): | 4.341 | Half-life (T1/2): | 0.026 |
——————————————————————————————————————————
ADMET: Toxicity
| hERG Blockers: | 0.228 | Human Hepatotoxicity (H-HT): | 0.006 |
| Drug-inuced Liver Injury (DILI): | 0.087 | AMES Toxicity: | 0.012 |
| Rat Oral Acute Toxicity: | 0.021 | Maximum Recommended Daily Dose: | 0.048 |
| Skin Sensitization: | 0.976 | Carcinogencity: | 0.036 |
| Eye Corrosion: | 0.996 | Eye Irritation: | 0.938 |
| Respiratory Toxicity: | 0.229 |
——————————————————————————————————————————
*Note: the ADMET properties was calculated by ADMETlab 2.0. Reference: PMID: 33893803.
Similar Compounds*
Compounds similar to EMNPD with top10 similarity:
| Similar NPs | Similar Drugs | ||||||
|---|---|---|---|---|---|---|---|
| NPs ID | NPs 2D Structure | Similarity Score | TTD ID | Drug 2D Structure | Similarity Score | ||
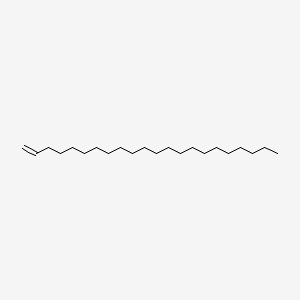
| ENC000755 |  |
0.914 | D00AOJ | 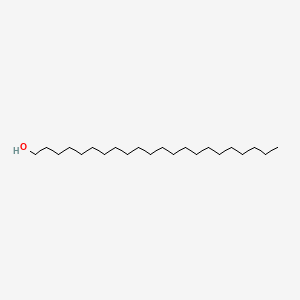 |
0.747 | ||
| ENC000589 | 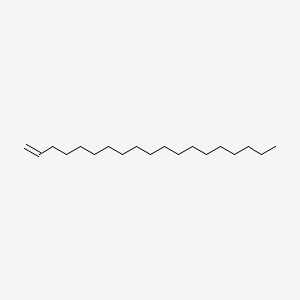 |
0.859 | D07ILQ | 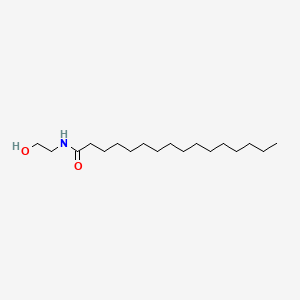 |
0.531 | ||
| ENC000591 |  |
0.842 | D00FGR |  |
0.495 | ||
| ENC000283 |  |
0.813 | D0Z5SM |  |
0.475 | ||
| ENC000723 |  |
0.812 | D00STJ |  |
0.459 | ||
| ENC000431 |  |
0.812 | D0O1PH |  |
0.444 | ||
| ENC000430 | 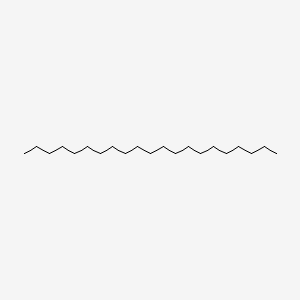 |
0.812 | D05ATI | 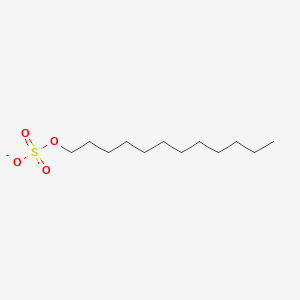 |
0.405 | ||
| ENC001176 | 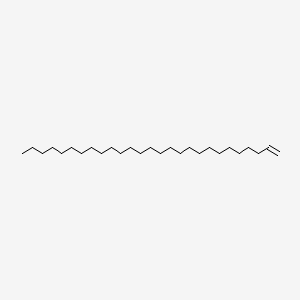 |
0.810 | D0Z5BC | 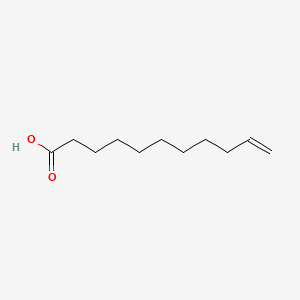 |
0.351 | ||
| ENC000923 | 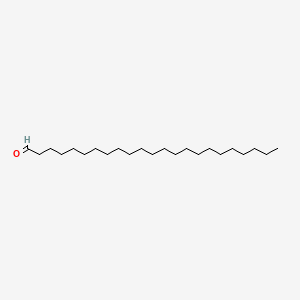 |
0.787 | D0T9TJ | 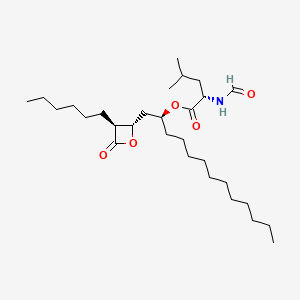 |
0.339 | ||
| ENC000432 |  |
0.778 | D0P1RL | 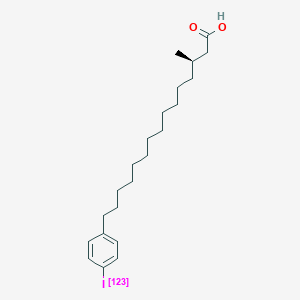 |
0.311 | ||