NPs Basic Information

|
Name |
1-Pentadecanol
|
| Molecular Formula | C15H32O | |
| IUPAC Name* |
pentadecan-1-ol
|
|
| SMILES |
CCCCCCCCCCCCCCCO
|
|
| InChI |
InChI=1S/C15H32O/c1-2-3-4-5-6-7-8-9-10-11-12-13-14-15-16/h16H,2-15H2,1H3
|
|
| InChIKey |
REIUXOLGHVXAEO-UHFFFAOYSA-N
|
|
| Synonyms |
1-PENTADECANOL; pentadecan-1-ol; 629-76-5; Pentadecyl alcohol; Pentadecanol; n-Pentadecanol; n-1-Pentadecanol; Neodol 5; MFCD00004759; 333QVA4G2Q; CHEBI:77468; NSC-66446; DSSTox_CID_7270; DSSTox_RID_78381; DSSTox_GSID_27270; 67762-25-8; n-pentadecyl alcohol; CAS-629-76-5; Alfol 15; 31389-11-4; UNII-333QVA4G2Q; n-pPentadecanol; Pentadecanol-(1); EINECS 211-107-9; NSC 66446; 1-Pentadecanol, 99%; AI3-33881; SCHEMBL29548; CHEMBL26561; DTXSID0027270; AMY5936; PENTADECYL ALCOHOL [INCI]; 1-pentadecanol (ACD/Name 4.0); NSC66446; EINECS 267-006-5; Tox21_201699; Tox21_300553; LMFA05000194; STL453722; ZINC38141455; AKOS009031435; CS-W004295; HY-W004295; Pentadecanol,95%; n-Pentadecyl alcohol; NCGC00164169-01; NCGC00164169-02; NCGC00164169-03; NCGC00254478-01; NCGC00259248-01; BP-30139; SY049687; FT-0608196; P0036; EN300-20042; H10901; A868289; Q27146999; F0001-1704; 1991DA79-7140-48B0-9F03-1E3D88AC4F28
|
|
| CAS | 629-76-5 | |
| PubChem CID | 12397 | |
| ChEMBL ID | CHEMBL26561 |
*Note: the IUPAC Name was collected from PubChem.
Chemical Classification: |
|
|
|---|
——————————————————————————————————————————
NPs Species Source
| Endophyte ID | Endophyte Name | Family | Genus | Taxonomy ID | GenBank ID | Closest GenBank ID | Reference | |
|---|---|---|---|---|---|---|---|---|
| Endophyte ID | Endophyte Name | Family | Genus | Taxonomy ID | GenBank ID | Closest GenBank ID | Reference |
NPs Biological Activity
| Bioactivity Name | Target ID | Target Name | Target Type | Target Organism | Target Organism ID | Potency of Bioactivity | Activity Type | Value | Unit | Endophyte ID | Endophyte Name | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Bioactivity Name | Target ID | Target Name | Target Type | Target Organism | Target Organism ID | Potency of Bioactivity | Activity Type | Value | Unit | Endophyte ID | Endophyte Name |
NPs Physi-Chem Properties
| Molecular Weight: | 228.41 | ALogp: | 6.8 |
| HBD: | 1 | HBA: | 1 |
| Rotatable Bonds: | 13 | Lipinski's rule of five: | Rejected |
| Polar Surface Area: | 20.2 | Aromatic Rings: | 0 |
| Heavy Atoms: | 16 | QED Weighted: | 0.419 |
——————————————————————————————————————————
NPs ADMET Properties*
ADMET: Absorption
| Caco-2 Permeability: | -4.674 | MDCK Permeability: | 0.00001670 |
| Pgp-inhibitor: | 0.002 | Pgp-substrate: | 0.003 |
| Human Intestinal Absorption (HIA): | 0.004 | 20% Bioavailability (F20%): | 0.174 |
| 30% Bioavailability (F30%): | 0.991 |
——————————————————————————————————————————
ADMET: Distribution
| Blood-Brain-Barrier Penetration (BBB): | 0.218 | Plasma Protein Binding (PPB): | 97.36% |
| Volume Distribution (VD): | 2.52 | Fu: | 1.72% |
——————————————————————————————————————————
ADMET: Metabolism
| CYP1A2-inhibitor: | 0.547 | CYP1A2-substrate: | 0.2 |
| CYP2C19-inhibitor: | 0.393 | CYP2C19-substrate: | 0.058 |
| CYP2C9-inhibitor: | 0.157 | CYP2C9-substrate: | 0.933 |
| CYP2D6-inhibitor: | 0.021 | CYP2D6-substrate: | 0.053 |
| CYP3A4-inhibitor: | 0.155 | CYP3A4-substrate: | 0.048 |
——————————————————————————————————————————
ADMET: Excretion
| Clearance (CL): | 5.95 | Half-life (T1/2): | 0.179 |
——————————————————————————————————————————
ADMET: Toxicity
| hERG Blockers: | 0.199 | Human Hepatotoxicity (H-HT): | 0.012 |
| Drug-inuced Liver Injury (DILI): | 0.051 | AMES Toxicity: | 0.006 |
| Rat Oral Acute Toxicity: | 0.024 | Maximum Recommended Daily Dose: | 0.013 |
| Skin Sensitization: | 0.95 | Carcinogencity: | 0.048 |
| Eye Corrosion: | 0.993 | Eye Irritation: | 0.942 |
| Respiratory Toxicity: | 0.501 |
——————————————————————————————————————————
*Note: the ADMET properties was calculated by ADMETlab 2.0. Reference: PMID: 33893803.
Similar Compounds*
Compounds similar to EMNPD with top10 similarity:
| Similar NPs | Similar Drugs | ||||||
|---|---|---|---|---|---|---|---|
| NPs ID | NPs 2D Structure | Similarity Score | TTD ID | Drug 2D Structure | Similarity Score | ||
| ENC000082 |  |
0.939 | D07ILQ | 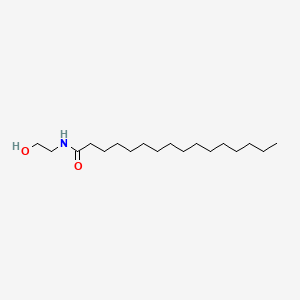 |
0.710 | ||
| ENC000486 |  |
0.885 | D00AOJ | 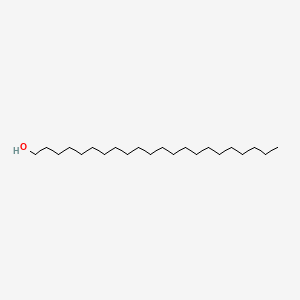 |
0.687 | ||
| ENC000284 |  |
0.836 | D0Z5SM |  |
0.613 | ||
| ENC000276 |  |
0.804 | D05ATI | 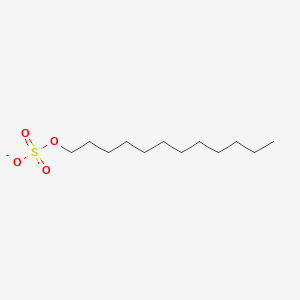 |
0.525 | ||
| ENC000379 |  |
0.804 | D0O1PH | 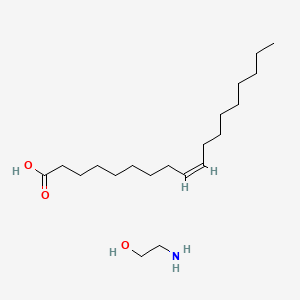 |
0.514 | ||
| ENC000745 |  |
0.793 | D00FGR | 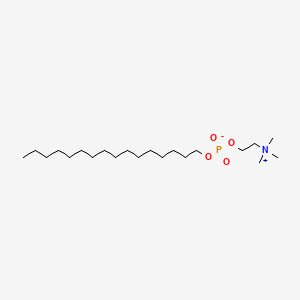 |
0.513 | ||
| ENC000050 |  |
0.764 | D0P1RL | 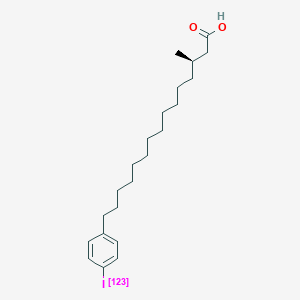 |
0.393 | ||
| ENC000380 |  |
0.759 | D00STJ | 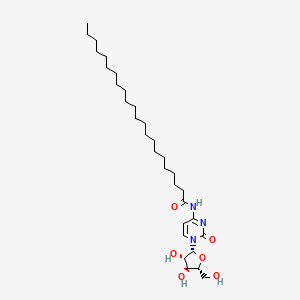 |
0.368 | ||
| ENC000557 |  |
0.759 | D0MM8N |  |
0.366 | ||
| ENC000427 |  |
0.759 | D05QNO | 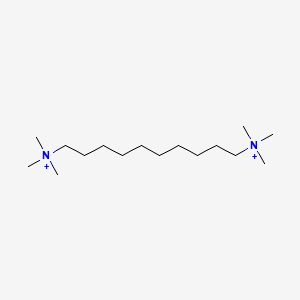 |
0.362 | ||