NPs Basic Information

|
Name |
4,6-Di-tert-butyl-m-cresol
|
| Molecular Formula | C15H24O | |
| IUPAC Name* |
2,4-ditert-butyl-5-methylphenol
|
|
| SMILES |
CC1=CC(=C(C=C1C(C)(C)C)C(C)(C)C)O
|
|
| InChI |
InChI=1S/C15H24O/c1-10-8-13(16)12(15(5,6)7)9-11(10)14(2,3)4/h8-9,16H,1-7H3
|
|
| InChIKey |
WYSSJDOPILWQDC-UHFFFAOYSA-N
|
|
| Synonyms |
4,6-Di-tert-butyl-m-cresol; 497-39-2; 2,4-Di-tert-butyl-5-methylphenol; DBMC; 4,6-DI-TERT-BUTYL-3-METHYLPHENOL; 4,6-Di-t-butyl-m-cresol; 3-Methyl-4,6-di-tert-butylphenol; 2,4-Di-t-butyl-5-methylphenol; m-Cresol, 4,6-di-tert-butyl-; Phenol, 2,4-bis(1,1-dimethylethyl)-5-methyl-; 2,4-Ditert-butyl-5-methylphenol; 2,4-Bis(1,1-dimethylethyl)-5-methylphenol; TMY866FL30; 2,4-Di-tert-butyl-5-hydroxytoluene; Di-tert-butyl-m-cresol; HSDB 5669; EINECS 207-847-7; BRN 2094885; UNII-TMY866FL30; AI3-15481; DBMC [MI]; DSSTox_CID_21502; DSSTox_RID_79753; DSSTox_GSID_41502; 4-06-00-03510 (Beilstein Handbook Reference); SCHEMBL824520; CHEMBL3183034; DTXSID1041502; ZINC2040443; Tox21_301524; 2,4-Ditert-butyl-5-methylphenol #; MFCD00059208; AKOS015890396; NCGC00255488-01; CAS-497-39-2; DB-051683; CS-0153302; D1432; FT-0632231; 2,4-bis-(1,1-dimethylethyl)-5-methyl-phenol; 5-Methyl-2,4-bis(2-methyl-2-propanyl)phenol; D89855; 4,6-DI-TERT-BUTYL-3-METHYLPHENOL [HSDB]; A827834; W-109082; Q27290032
|
|
| CAS | 497-39-2 | |
| PubChem CID | 10346 | |
| ChEMBL ID | CHEMBL3183034 |
*Note: the IUPAC Name was collected from PubChem.
Chemical Classification: |
|
|
|---|
——————————————————————————————————————————
NPs Species Source
| Endophyte ID | Endophyte Name | Family | Genus | Taxonomy ID | GenBank ID | Closest GenBank ID | Reference | |
|---|---|---|---|---|---|---|---|---|
| Endophyte ID | Endophyte Name | Family | Genus | Taxonomy ID | GenBank ID | Closest GenBank ID | Reference |
NPs Biological Activity
| Bioactivity Name | Target ID | Target Name | Target Type | Target Organism | Target Organism ID | Potency of Bioactivity | Activity Type | Value | Unit | Endophyte ID | Endophyte Name | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Bioactivity Name | Target ID | Target Name | Target Type | Target Organism | Target Organism ID | Potency of Bioactivity | Activity Type | Value | Unit | Endophyte ID | Endophyte Name |
NPs Physi-Chem Properties
| Molecular Weight: | 220.35 | ALogp: | 5.3 |
| HBD: | 1 | HBA: | 1 |
| Rotatable Bonds: | 2 | Lipinski's rule of five: | Accepted |
| Polar Surface Area: | 20.2 | Aromatic Rings: | 1 |
| Heavy Atoms: | 16 | QED Weighted: | 0.663 |
——————————————————————————————————————————
NPs ADMET Properties*
ADMET: Absorption
| Caco-2 Permeability: | -5.318 | MDCK Permeability: | 0.00000972 |
| Pgp-inhibitor: | 0.334 | Pgp-substrate: | 0.006 |
| Human Intestinal Absorption (HIA): | 0.935 | 20% Bioavailability (F20%): | 0.998 |
| 30% Bioavailability (F30%): | 0.997 |
——————————————————————————————————————————
ADMET: Distribution
| Blood-Brain-Barrier Penetration (BBB): | 0.411 | Plasma Protein Binding (PPB): | 98.85% |
| Volume Distribution (VD): | 5.222 | Fu: | 4.21% |
——————————————————————————————————————————
ADMET: Metabolism
| CYP1A2-inhibitor: | 0.862 | CYP1A2-substrate: | 0.945 |
| CYP2C19-inhibitor: | 0.613 | CYP2C19-substrate: | 0.835 |
| CYP2C9-inhibitor: | 0.499 | CYP2C9-substrate: | 0.858 |
| CYP2D6-inhibitor: | 0.874 | CYP2D6-substrate: | 0.9 |
| CYP3A4-inhibitor: | 0.519 | CYP3A4-substrate: | 0.664 |
——————————————————————————————————————————
ADMET: Excretion
| Clearance (CL): | 6.339 | Half-life (T1/2): | 0.263 |
——————————————————————————————————————————
ADMET: Toxicity
| hERG Blockers: | 0.016 | Human Hepatotoxicity (H-HT): | 0.04 |
| Drug-inuced Liver Injury (DILI): | 0.044 | AMES Toxicity: | 0.01 |
| Rat Oral Acute Toxicity: | 0.094 | Maximum Recommended Daily Dose: | 0.916 |
| Skin Sensitization: | 0.49 | Carcinogencity: | 0.03 |
| Eye Corrosion: | 0.962 | Eye Irritation: | 0.978 |
| Respiratory Toxicity: | 0.811 |
——————————————————————————————————————————
*Note: the ADMET properties was calculated by ADMETlab 2.0. Reference: PMID: 33893803.
Similar Compounds*
Compounds similar to EMNPD with top10 similarity:
| Similar NPs | Similar Drugs | ||||||
|---|---|---|---|---|---|---|---|
| NPs ID | NPs 2D Structure | Similarity Score | TTD ID | Drug 2D Structure | Similarity Score | ||
| ENC000079 |  |
0.702 | D0W7WC |  |
0.405 | ||
| ENC000610 |  |
0.667 | D09EBS |  |
0.319 | ||
| ENC000725 |  |
0.600 | D0H2DQ | 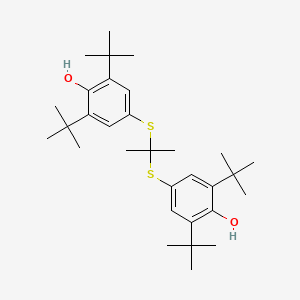 |
0.316 | ||
| ENC000708 |  |
0.566 | D0Y4DY |  |
0.290 | ||
| ENC005113 | 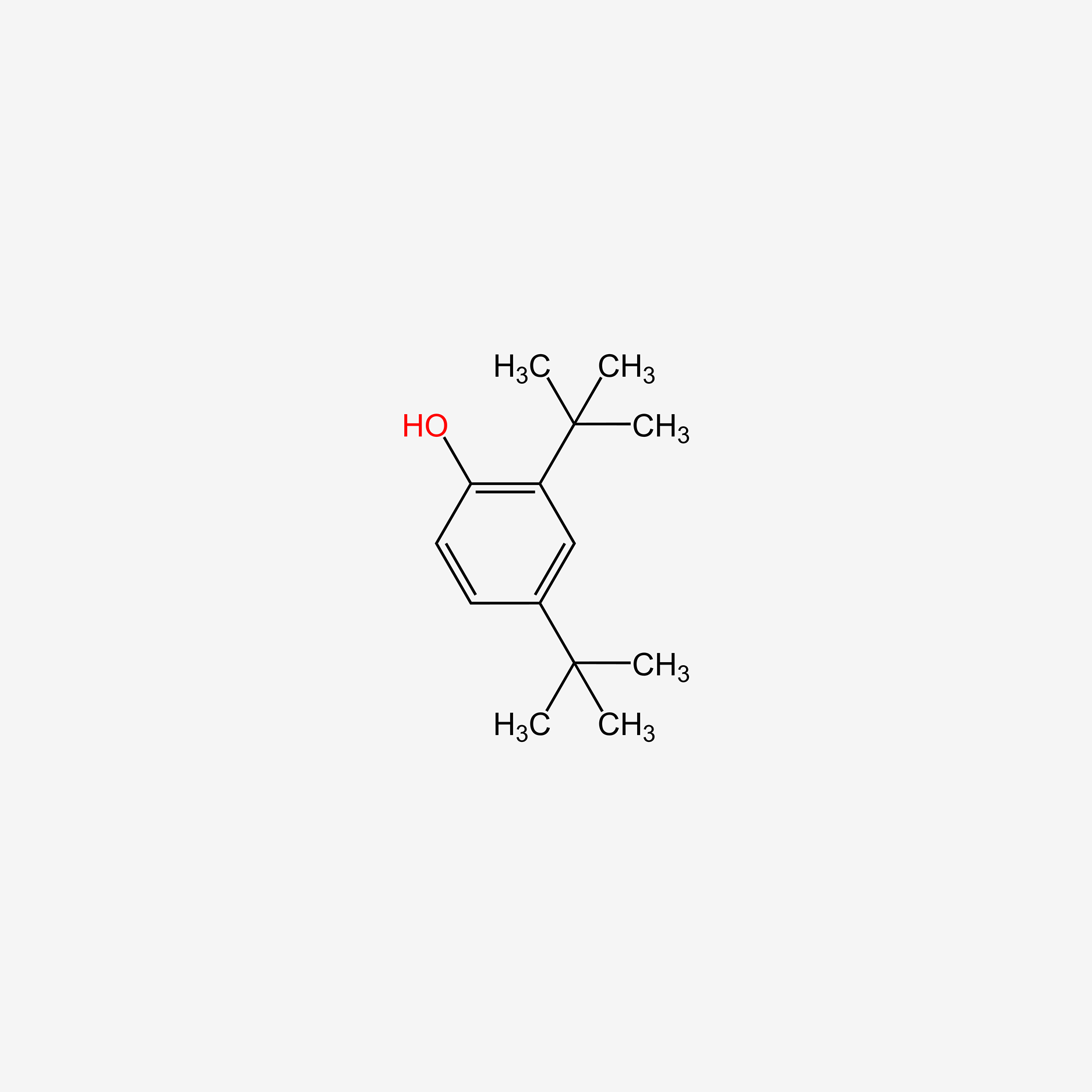 |
0.560 | D00NJL |  |
0.284 | ||
| ENC000185 | 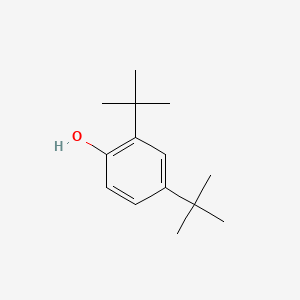 |
0.560 | D01JFT | 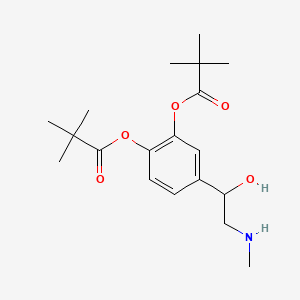 |
0.265 | ||
| ENC000744 | 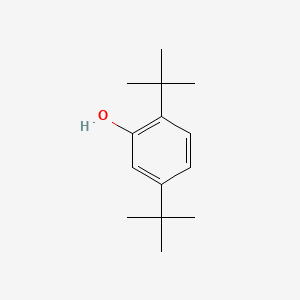 |
0.560 | D0M8RC | 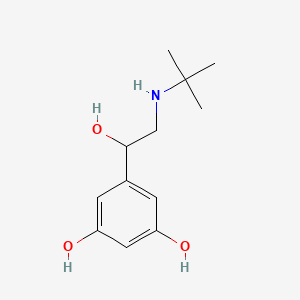 |
0.242 | ||
| ENC000611 | 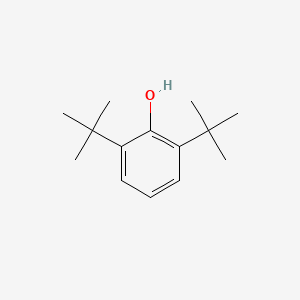 |
0.529 | D0X5NX |  |
0.235 | ||
| ENC000658 |  |
0.492 | D06GIP |  |
0.232 | ||
| ENC001398 |  |
0.491 | D0K5CB | 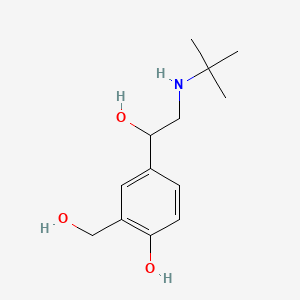 |
0.232 | ||