NPs Basic Information

|
Name |
(S)-(-)-5-(hydroxymethyl)-2-(2′,6′,6′-trimethyltetrahydro-2H-pyran-2-yl)phenol
|
| Molecular Formula | C16H26O4 | |
| IUPAC Name* |
8-hydroxy-2-(1-hydroxyheptyl)-2,3,4,6,7,8-hexahydrochromen-5-one
|
|
| SMILES |
CCCCCCC(O)C1CCC2=C(O1)C(O)CCC2=O
|
|
| InChI |
InChI=1S/C16H26O4/c1-2-3-4-5-6-13(18)15-10-7-11-12(17)8-9-14(19)16(11)20-15/h13-15,18-19H,2-10H2,1H3/t13-,14+,15-/m0/s1
|
|
| InChIKey |
INHVGPIPHZJQOP-ZNMIVQPWSA-N
|
|
| Synonyms |
NA
|
|
| CAS | NA | |
| PubChem CID | NA | |
| ChEMBL ID | NA |
*Note: the IUPAC Name was calculated by STOUT. Reference: PMID:33906675.
Chemical Classification: |
|
|
|---|
——————————————————————————————————————————
NPs Species Source
| Endophyte ID | Endophyte Name | Family | Genus | Taxonomy ID | GenBank ID | Closest GenBank ID | Reference | |
|---|---|---|---|---|---|---|---|---|
| Endophyte ID | Endophyte Name | Family | Genus | Taxonomy ID | GenBank ID | Closest GenBank ID | Reference |
NPs Biological Activity
| Bioactivity Name | Target ID | Target Name | Target Type | Target Organism | Target Organism ID | Potency of Bioactivity | Activity Type | Value | Unit | Endophyte ID | Endophyte Name | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Bioactivity Name | Target ID | Target Name | Target Type | Target Organism | Target Organism ID | Potency of Bioactivity | Activity Type | Value | Unit | Endophyte ID | Endophyte Name |
NPs Physi-Chem Properties
| Molecular Weight: | 282.38 | ALogp: | 2.5 |
| HBD: | 2 | HBA: | 4 |
| Rotatable Bonds: | 6 | Lipinski's rule of five: | Accepted |
| Polar Surface Area: | 66.8 | Aromatic Rings: | 2 |
| Heavy Atoms: | 20 | QED Weighted: | 0.734 |
——————————————————————————————————————————
NPs ADMET Properties*
ADMET: Absorption
| Caco-2 Permeability: | -4.688 | MDCK Permeability: | 0.00002700 |
| Pgp-inhibitor: | 0.336 | Pgp-substrate: | 0.071 |
| Human Intestinal Absorption (HIA): | 0.097 | 20% Bioavailability (F20%): | 0.976 |
| 30% Bioavailability (F30%): | 0.424 |
——————————————————————————————————————————
ADMET: Distribution
| Blood-Brain-Barrier Penetration (BBB): | 0.404 | Plasma Protein Binding (PPB): | 78.10% |
| Volume Distribution (VD): | 0.977 | Fu: | 14.30% |
——————————————————————————————————————————
ADMET: Metabolism
| CYP1A2-inhibitor: | 0.025 | CYP1A2-substrate: | 0.787 |
| CYP2C19-inhibitor: | 0.031 | CYP2C19-substrate: | 0.671 |
| CYP2C9-inhibitor: | 0.03 | CYP2C9-substrate: | 0.853 |
| CYP2D6-inhibitor: | 0.004 | CYP2D6-substrate: | 0.68 |
| CYP3A4-inhibitor: | 0.018 | CYP3A4-substrate: | 0.103 |
——————————————————————————————————————————
ADMET: Excretion
| Clearance (CL): | 13.235 | Half-life (T1/2): | 0.835 |
——————————————————————————————————————————
ADMET: Toxicity
| hERG Blockers: | 0.015 | Human Hepatotoxicity (H-HT): | 0.296 |
| Drug-inuced Liver Injury (DILI): | 0.293 | AMES Toxicity: | 0.051 |
| Rat Oral Acute Toxicity: | 0.939 | Maximum Recommended Daily Dose: | 0.256 |
| Skin Sensitization: | 0.056 | Carcinogencity: | 0.228 |
| Eye Corrosion: | 0.004 | Eye Irritation: | 0.07 |
| Respiratory Toxicity: | 0.032 |
——————————————————————————————————————————
*Note: the ADMET properties was calculated by ADMETlab 2.0. Reference: PMID: 33893803.
Similar Compounds*
Compounds similar to EMNPD with top10 similarity:
| Similar NPs | Similar Drugs | ||||||
|---|---|---|---|---|---|---|---|
| NPs ID | NPs 2D Structure | Similarity Score | TTD ID | Drug 2D Structure | Similarity Score | ||
 |
D0V0IX | 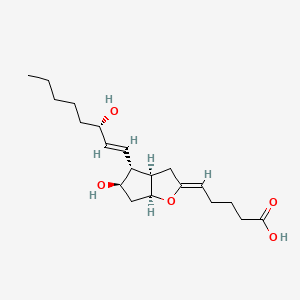 |
0.286 | ||||
 |
D0I4DQ | 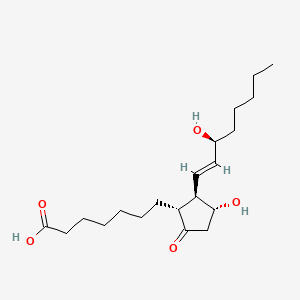 |
0.276 | ||||
 |
D0L7AS | 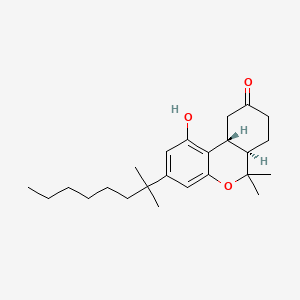 |
0.275 | ||||
 |
D0XN8C | 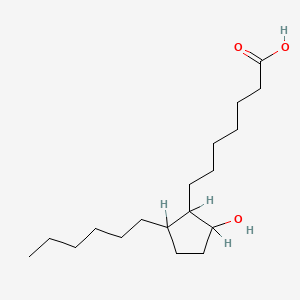 |
0.264 | ||||
 |
D06FEA |  |
0.263 | ||||
 |
D01WUA |  |
0.262 | ||||
 |
D00CTS |  |
0.238 | ||||
 |
D03ZJE |  |
0.237 | ||||
 |
D02AXG |  |
0.230 | ||||
 |
D0O1UZ |  |
0.227 | ||||