NPs Basic Information

|
Name |
Erucic Acid
|
| Molecular Formula | C22H42O2 | |
| IUPAC Name* |
(Z)-docos-13-enoic acid
|
|
| SMILES |
CCCCCCCC/C=C\CCCCCCCCCCCC(=O)O
|
|
| InChI |
InChI=1S/C22H42O2/c1-2-3-4-5-6-7-8-9-10-11-12-13-14-15-16-17-18-19-20-21-22(23)24/h9-10H,2-8,11-21H2,1H3,(H,23,24)/b10-9-
|
|
| InChIKey |
DPUOLQHDNGRHBS-KTKRTIGZSA-N
|
|
| Synonyms |
ERUCIC ACID; 112-86-7; (Z)-Docos-13-enoic acid; cis-13-Docosenoic acid; 13-Docosenoic acid, (Z)-; 13-cis-Docosenoic acid; Hystrene 2290; cis-erucic acid; 13-Docosenoic acid; 13Z-docosenoic acid; Prifrac 2990; (Z)-13-Docosenoic acid; (13Z)-13-Docosenoic acid; (13Z)-docos-13-enoic acid; .delta.13-cis-Docosenoic acid; CHEBI:28792; (13Z)-Docosenoic acid; 13-Docosenoic acid, (13Z)-; 13(Z)-Docosenoic Acid; C22:1n-9; MFCD00063188; NSC-6814; 075441GMF2; 13-Docosenoic acid, (13Z)-, dimer; (Z)-13-docosenoate; Z-13-Docosenoic acid; 63541-50-4; 1072-39-5; delta13-cis-Docosenoic acid; delta13:14-Docosenoic acid; HSDB 5015; Prifac 2990; NSC 6814; EINECS 204-011-3; NSC6814; Erucasaeure; AI3-18180; cis-eruic acid; UNII-075441GMF2; 13-docosenoate; cis-13-Docosenoate; Erucic acid, 80%; docos-13c-enoic acid; 22:1omega9; Erucic acid, >=99%; delta 13-cis-Docosenoate; delta.13-cis-Docosenoate; NOURACID RE 07; DSSTox_CID_6931; ERUCIC ACID [MI]; (Z)-Docos-13-enoicacid; (13Z)-13-Docosenoate; ERUCIC ACID [HSDB]; ERUCIC ACID [INCI]; SCHEMBL5987; DSSTox_RID_78259; DSSTox_GSID_26931; delta 13-cis-Docosenoic acid; delta.13-cis-Docosenoic acid; Docosenoic acid, 13-(Z)-; cis-Delta(13)-docosenoic acid; JARIC 22:1; CHEMBL1173380; DTXSID8026931; Erucic acid, analytical standard; (13Z)-13-Docosenoic acid #; HMS3649F15; HY-N7109; ZINC8220981; Tox21_200242; BDBM50463967; LMFA01030089; AKOS015961814; Erucic acid, >=99% (capillary GC); Erucic acid, technical, ~90% (GC); 13-docosenoic acid (ACD/Name 4.0); CCG-267904; NCGC00166073-01; NCGC00166073-02; NCGC00257796-01; CAS-112-86-7; FA(22:1(13Z)); CS-0014094; CS-0203830; D0965; S5383; C08316; F17010; Q413531; SR-01000946653; SR-01000946653-1; W-108635; 22:1(N-9); 084C86E4-C0FF-45E5-9F1D-09913C9193BE
|
|
| CAS | 112-86-7 | |
| PubChem CID | 5281116 | |
| ChEMBL ID | CHEMBL1173380 |
*Note: the IUPAC Name was collected from PubChem.
Chemical Classification: |
|
|
|---|
——————————————————————————————————————————
NPs Species Source
| Endophyte ID | Endophyte Name | Family | Genus | Taxonomy ID | GenBank ID | Closest GenBank ID | Reference | |
|---|---|---|---|---|---|---|---|---|
| Endophyte ID | Endophyte Name | Family | Genus | Taxonomy ID | GenBank ID | Closest GenBank ID | Reference |
NPs Biological Activity
| Bioactivity Name | Target ID | Target Name | Target Type | Target Organism | Target Organism ID | Potency of Bioactivity | Activity Type | Value | Unit | Endophyte ID | Endophyte Name | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Bioactivity Name | Target ID | Target Name | Target Type | Target Organism | Target Organism ID | Potency of Bioactivity | Activity Type | Value | Unit | Endophyte ID | Endophyte Name |
NPs Physi-Chem Properties
| Molecular Weight: | 338.6 | ALogp: | 8.7 |
| HBD: | 1 | HBA: | 2 |
| Rotatable Bonds: | 19 | Lipinski's rule of five: | Rejected |
| Polar Surface Area: | 37.3 | Aromatic Rings: | 0 |
| Heavy Atoms: | 24 | QED Weighted: | 0.19 |
——————————————————————————————————————————
NPs ADMET Properties*
ADMET: Absorption
| Caco-2 Permeability: | -5.092 | MDCK Permeability: | 0.00002760 |
| Pgp-inhibitor: | 0 | Pgp-substrate: | 0 |
| Human Intestinal Absorption (HIA): | 0.047 | 20% Bioavailability (F20%): | 0.933 |
| 30% Bioavailability (F30%): | 0.997 |
——————————————————————————————————————————
ADMET: Distribution
| Blood-Brain-Barrier Penetration (BBB): | 0.009 | Plasma Protein Binding (PPB): | 100.10% |
| Volume Distribution (VD): | 1.263 | Fu: | 0.45% |
——————————————————————————————————————————
ADMET: Metabolism
| CYP1A2-inhibitor: | 0.174 | CYP1A2-substrate: | 0.182 |
| CYP2C19-inhibitor: | 0.246 | CYP2C19-substrate: | 0.062 |
| CYP2C9-inhibitor: | 0.102 | CYP2C9-substrate: | 0.994 |
| CYP2D6-inhibitor: | 0.029 | CYP2D6-substrate: | 0.113 |
| CYP3A4-inhibitor: | 0.109 | CYP3A4-substrate: | 0.014 |
——————————————————————————————————————————
ADMET: Excretion
| Clearance (CL): | 2.689 | Half-life (T1/2): | 0.704 |
——————————————————————————————————————————
ADMET: Toxicity
| hERG Blockers: | 0.113 | Human Hepatotoxicity (H-HT): | 0.04 |
| Drug-inuced Liver Injury (DILI): | 0.019 | AMES Toxicity: | 0.012 |
| Rat Oral Acute Toxicity: | 0.024 | Maximum Recommended Daily Dose: | 0.023 |
| Skin Sensitization: | 0.956 | Carcinogencity: | 0.054 |
| Eye Corrosion: | 0.923 | Eye Irritation: | 0.966 |
| Respiratory Toxicity: | 0.898 |
——————————————————————————————————————————
*Note: the ADMET properties was calculated by ADMETlab 2.0. Reference: PMID: 33893803.
Similar Compounds*
Compounds similar to EMNPD with top10 similarity:
| Similar NPs | Similar Drugs | ||||||
|---|---|---|---|---|---|---|---|
| NPs ID | NPs 2D Structure | Similarity Score | TTD ID | Drug 2D Structure | Similarity Score | ||
| ENC001593 |  |
0.913 | D0O1PH | 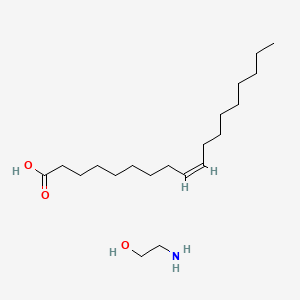 |
0.776 | ||
| ENC001710 |  |
0.865 | D07ILQ | 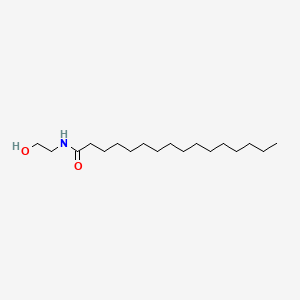 |
0.613 | ||
| ENC001678 |  |
0.831 | D0O1TC | 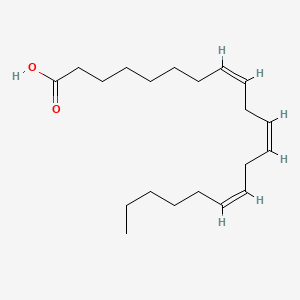 |
0.553 | ||
| ENC001592 |  |
0.826 | D00AOJ | 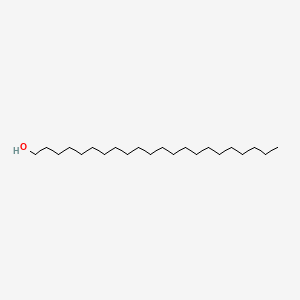 |
0.545 | ||
| ENC001100 |  |
0.826 | D0UE9X |  |
0.482 | ||
| ENC001555 |  |
0.826 | D00FGR | 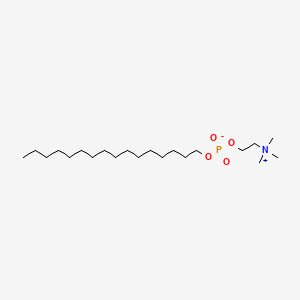 |
0.469 | ||
| ENC001419 |  |
0.826 | D0Z5SM |  |
0.464 | ||
| ENC001591 |  |
0.826 | D0Z5BC | 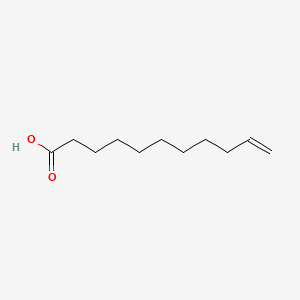 |
0.419 | ||
| ENC001775 |  |
0.800 | D0OR6A |  |
0.415 | ||
| ENC000110 |  |
0.775 | D00STJ | 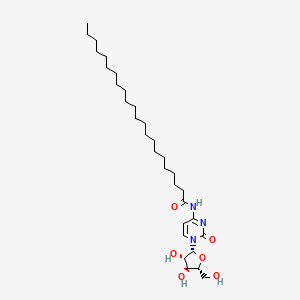 |
0.408 | ||