NPs Basic Information

|
Name |
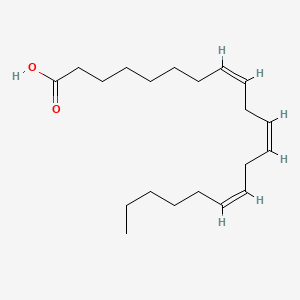
2,3-Nonadiene
|
| Molecular Formula | C9H16 | |
| IUPAC Name* |
NA
|
|
| SMILES |
CCCCCC=C=CC
|
|
| InChI |
InChI=1S/C9H16/c1-3-5-7-9-8-6-4-2/h3,7H,4,6,8-9H2,1-2H3
|
|
| InChIKey |
QPYQHAPIBCQICN-UHFFFAOYSA-N
|
|
| Synonyms |
2,3-Nonadiene; 22433-34-7; DTXSID60338477
|
|
| CAS | 22433-34-7 | |
| PubChem CID | 549311 | |
| ChEMBL ID | NA |
*Note: the IUPAC Name was collected from PubChem.
Chemical Classification: |
|
|
|---|
——————————————————————————————————————————
NPs Physi-Chem Properties
| Molecular Weight: | 124.22 | ALogp: | 3.3 |
| HBD: | 0 | HBA: | 0 |
| Rotatable Bonds: | 4 | Lipinski's rule of five: | Accepted |
| Polar Surface Area: | 0.0 | Aromatic Rings: | 0 |
| Heavy Atoms: | 9 | QED Weighted: | 0.391 |
——————————————————————————————————————————
NPs ADMET Properties*
ADMET: Absorption
| Caco-2 Permeability: | -4.487 | MDCK Permeability: | 0.00001030 |
| Pgp-inhibitor: | 0.101 | Pgp-substrate: | 0.085 |
| Human Intestinal Absorption (HIA): | 0.003 | 20% Bioavailability (F20%): | 0.005 |
| 30% Bioavailability (F30%): | 0.068 |
——————————————————————————————————————————
ADMET: Distribution
| Blood-Brain-Barrier Penetration (BBB): | 0.979 | Plasma Protein Binding (PPB): | 89.53% |
| Volume Distribution (VD): | 3.446 | Fu: | 15.42% |
——————————————————————————————————————————
ADMET: Metabolism
| CYP1A2-inhibitor: | 0.484 | CYP1A2-substrate: | 0.717 |
| CYP2C19-inhibitor: | 0.034 | CYP2C19-substrate: | 0.908 |
| CYP2C9-inhibitor: | 0.032 | CYP2C9-substrate: | 0.67 |
| CYP2D6-inhibitor: | 0.002 | CYP2D6-substrate: | 0.048 |
| CYP3A4-inhibitor: | 0.034 | CYP3A4-substrate: | 0.212 |
——————————————————————————————————————————
ADMET: Excretion
| Clearance (CL): | 7.204 | Half-life (T1/2): | 0.702 |
——————————————————————————————————————————
ADMET: Toxicity
| hERG Blockers: | 0.042 | Human Hepatotoxicity (H-HT): | 0.505 |
| Drug-inuced Liver Injury (DILI): | 0.043 | AMES Toxicity: | 0.008 |
| Rat Oral Acute Toxicity: | 0.043 | Maximum Recommended Daily Dose: | 0.134 |
| Skin Sensitization: | 0.967 | Carcinogencity: | 0.597 |
| Eye Corrosion: | 0.992 | Eye Irritation: | 0.987 |
| Respiratory Toxicity: | 0.893 |