NPs Basic Information

|
Name |
Methyl pentadecanoate
|
| Molecular Formula | C16H32O2 | |
| IUPAC Name* |
methyl pentadecanoate
|
|
| SMILES |
CCCCCCCCCCCCCCC(=O)OC
|
|
| InChI |
InChI=1S/C16H32O2/c1-3-4-5-6-7-8-9-10-11-12-13-14-15-16(17)18-2/h3-15H2,1-2H3
|
|
| InChIKey |
XIUXKAZJZFLLDQ-UHFFFAOYSA-N
|
|
| Synonyms |
METHYL PENTADECANOATE; 7132-64-1; Pentadecanoic acid, methyl ester; Pentadecanoic acid methyl ester; Methyl n-pentadecanoate; n-Pentadecanoic acid methyl ester; MFCD00008989; 8K0ZV6FAIZ; pentadecanoic acid-methyl ester; 68937-84-8; UNII-8K0ZV6FAIZ; EINECS 230-430-6; EINECS 273-095-1; pentadecanoic acid methyl; AI3-36452; Methyl pentadecanoate, 98%; SCHEMBL246530; CHEMBL1900809; DTXSID4040769; CDAA-251015M; CHEBI:142657; Methyl pentadecanoate, >=98.5%; Pentanedecanoic acid, methyl ester; STL454743; ZINC38141470; AKOS002676182; CS-W004289; HY-W004289; NCGC00164331-01; AS-57591; Methyl pentadecanoate, analytical standard; SY051773; DB-055514; FT-0636359; P0869; H10934; A914605; Q27270652; 93612FD5-1EFF-4869-9B03-8B8BAD63EE28
|
|
| CAS | 7132-64-1 | |
| PubChem CID | 23518 | |
| ChEMBL ID | CHEMBL1900809 |
*Note: the IUPAC Name was collected from PubChem.
Chemical Classification: |
|
|
|---|
——————————————————————————————————————————
NPs Species Source
| Endophyte ID | Endophyte Name | Family | Genus | Taxonomy ID | GenBank ID | Closest GenBank ID | Reference | |
|---|---|---|---|---|---|---|---|---|
| Endophyte ID | Endophyte Name | Family | Genus | Taxonomy ID | GenBank ID | Closest GenBank ID | Reference |
NPs Biological Activity
| Bioactivity Name | Target ID | Target Name | Target Type | Target Organism | Target Organism ID | Potency of Bioactivity | Activity Type | Value | Unit | Endophyte ID | Endophyte Name | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Bioactivity Name | Target ID | Target Name | Target Type | Target Organism | Target Organism ID | Potency of Bioactivity | Activity Type | Value | Unit | Endophyte ID | Endophyte Name |
NPs Physi-Chem Properties
| Molecular Weight: | 256.42 | ALogp: | 7.4 |
| HBD: | 0 | HBA: | 2 |
| Rotatable Bonds: | 14 | Lipinski's rule of five: | Rejected |
| Polar Surface Area: | 26.3 | Aromatic Rings: | 0 |
| Heavy Atoms: | 18 | QED Weighted: | 0.32 |
——————————————————————————————————————————
NPs ADMET Properties*
ADMET: Absorption
| Caco-2 Permeability: | -4.728 | MDCK Permeability: | 0.00001720 |
| Pgp-inhibitor: | 0.071 | Pgp-substrate: | 0.002 |
| Human Intestinal Absorption (HIA): | 0.002 | 20% Bioavailability (F20%): | 0.983 |
| 30% Bioavailability (F30%): | 0.995 |
——————————————————————————————————————————
ADMET: Distribution
| Blood-Brain-Barrier Penetration (BBB): | 0.571 | Plasma Protein Binding (PPB): | 97.21% |
| Volume Distribution (VD): | 1.69 | Fu: | 1.63% |
——————————————————————————————————————————
ADMET: Metabolism
| CYP1A2-inhibitor: | 0.77 | CYP1A2-substrate: | 0.217 |
| CYP2C19-inhibitor: | 0.519 | CYP2C19-substrate: | 0.134 |
| CYP2C9-inhibitor: | 0.284 | CYP2C9-substrate: | 0.937 |
| CYP2D6-inhibitor: | 0.101 | CYP2D6-substrate: | 0.073 |
| CYP3A4-inhibitor: | 0.4 | CYP3A4-substrate: | 0.073 |
——————————————————————————————————————————
ADMET: Excretion
| Clearance (CL): | 5.197 | Half-life (T1/2): | 0.337 |
——————————————————————————————————————————
ADMET: Toxicity
| hERG Blockers: | 0.203 | Human Hepatotoxicity (H-HT): | 0.027 |
| Drug-inuced Liver Injury (DILI): | 0.306 | AMES Toxicity: | 0.006 |
| Rat Oral Acute Toxicity: | 0.028 | Maximum Recommended Daily Dose: | 0.018 |
| Skin Sensitization: | 0.952 | Carcinogencity: | 0.072 |
| Eye Corrosion: | 0.954 | Eye Irritation: | 0.972 |
| Respiratory Toxicity: | 0.904 |
——————————————————————————————————————————
*Note: the ADMET properties was calculated by ADMETlab 2.0. Reference: PMID: 33893803.
Similar Compounds*
Compounds similar to EMNPD with top10 similarity:
| Similar NPs | Similar Drugs | ||||||
|---|---|---|---|---|---|---|---|
| NPs ID | NPs 2D Structure | Similarity Score | TTD ID | Drug 2D Structure | Similarity Score | ||
| ENC000271 |  |
0.944 | D07ILQ | 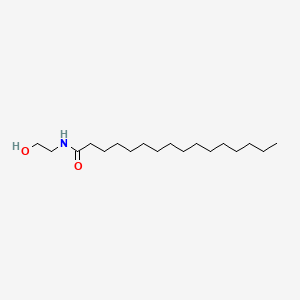 |
0.632 | ||
| ENC000604 |  |
0.941 | D0Z5SM |  |
0.615 | ||
| ENC000496 |  |
0.895 | D05ATI | 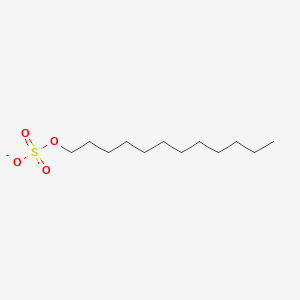 |
0.531 | ||
| ENC000495 |  |
0.882 | D0O1PH | 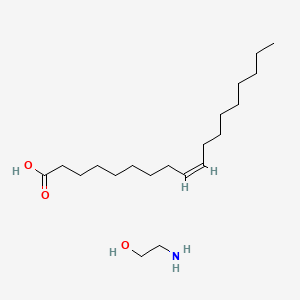 |
0.500 | ||
| ENC000280 |  |
0.850 | D00FGR | 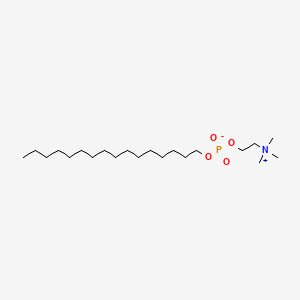 |
0.482 | ||
| ENC000260 |  |
0.824 | D00AOJ | 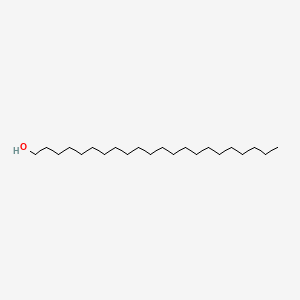 |
0.475 | ||
| ENC000497 |  |
0.810 | D00MLW | 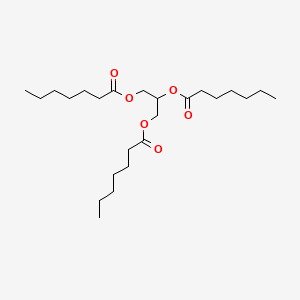 |
0.406 | ||
| ENC001160 |  |
0.783 | D0P1RL | 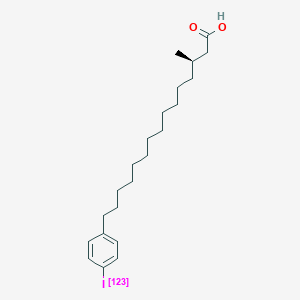 |
0.402 | ||
| ENC000474 |  |
0.773 | D0XN8C | 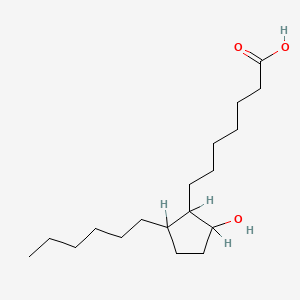 |
0.392 | ||
| ENC000419 | 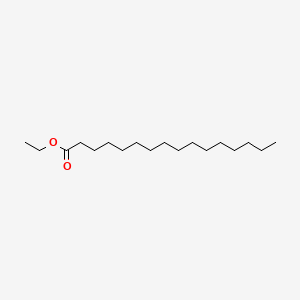 |
0.770 | D0T9TJ | 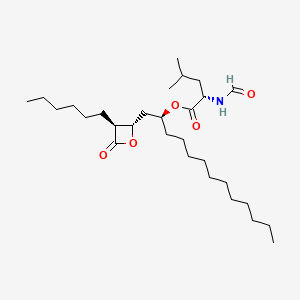 |
0.380 | ||