NPs Basic Information

|
Name |
1-Iodooctadecane
|
| Molecular Formula | C18H37I | |
| IUPAC Name* |
1-iodooctadecane
|
|
| SMILES |
CCCCCCCCCCCCCCCCCCI
|
|
| InChI |
InChI=1S/C18H37I/c1-2-3-4-5-6-7-8-9-10-11-12-13-14-15-16-17-18-19/h2-18H2,1H3
|
|
| InChIKey |
ZNJOCVLVYVOUGB-UHFFFAOYSA-N
|
|
| Synonyms |
1-IODOOCTADECANE; Octadecyl iodide; 629-93-6; Stearyl iodide; Octadecane, 1-iodo-; n-Octadecyl iodide; K8J7D7A7XG; NSC-5544; iodooctadecane; octadecyliodide; Octadecyljodid; NSC 5544; EINECS 211-117-3; 1-Iodooctadecane, 95%; UNII-K8J7D7A7XG; SCHEMBL226201; DTXSID7060881; NSC5544; ZINC6920368; MFCD00001090; AKOS015839785; AS-56768; CS-0179367; FT-0607967; I0065; D91071; W-104944
|
|
| CAS | 629-93-6 | |
| PubChem CID | 12402 | |
| ChEMBL ID | NA |
*Note: the IUPAC Name was collected from PubChem.
Chemical Classification: |
|
|
|---|
——————————————————————————————————————————
NPs Species Source
| Endophyte ID | Endophyte Name | Family | Genus | Taxonomy ID | GenBank ID | Closest GenBank ID | Reference | |
|---|---|---|---|---|---|---|---|---|
| Endophyte ID | Endophyte Name | Family | Genus | Taxonomy ID | GenBank ID | Closest GenBank ID | Reference |
NPs Biological Activity
| Bioactivity Name | Target ID | Target Name | Target Type | Target Organism | Target Organism ID | Potency of Bioactivity | Activity Type | Value | Unit | Endophyte ID | Endophyte Name | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Bioactivity Name | Target ID | Target Name | Target Type | Target Organism | Target Organism ID | Potency of Bioactivity | Activity Type | Value | Unit | Endophyte ID | Endophyte Name |
NPs Physi-Chem Properties
| Molecular Weight: | 380.4 | ALogp: | 10.7 |
| HBD: | 0 | HBA: | 0 |
| Rotatable Bonds: | 16 | Lipinski's rule of five: | Rejected |
| Polar Surface Area: | 0.0 | Aromatic Rings: | 0 |
| Heavy Atoms: | 19 | QED Weighted: | 0.143 |
——————————————————————————————————————————
NPs ADMET Properties*
ADMET: Absorption
| Caco-2 Permeability: | -4.934 | MDCK Permeability: | 0.00000728 |
| Pgp-inhibitor: | 0 | Pgp-substrate: | 0 |
| Human Intestinal Absorption (HIA): | 0.002 | 20% Bioavailability (F20%): | 0.138 |
| 30% Bioavailability (F30%): | 0.993 |
——————————————————————————————————————————
ADMET: Distribution
| Blood-Brain-Barrier Penetration (BBB): | 0.032 | Plasma Protein Binding (PPB): | 98.84% |
| Volume Distribution (VD): | 3.696 | Fu: | 1.17% |
——————————————————————————————————————————
ADMET: Metabolism
| CYP1A2-inhibitor: | 0.147 | CYP1A2-substrate: | 0.18 |
| CYP2C19-inhibitor: | 0.3 | CYP2C19-substrate: | 0.059 |
| CYP2C9-inhibitor: | 0.065 | CYP2C9-substrate: | 0.949 |
| CYP2D6-inhibitor: | 0.237 | CYP2D6-substrate: | 0.069 |
| CYP3A4-inhibitor: | 0.18 | CYP3A4-substrate: | 0.039 |
——————————————————————————————————————————
ADMET: Excretion
| Clearance (CL): | 4.081 | Half-life (T1/2): | 0.04 |
——————————————————————————————————————————
ADMET: Toxicity
| hERG Blockers: | 0.257 | Human Hepatotoxicity (H-HT): | 0.039 |
| Drug-inuced Liver Injury (DILI): | 0.609 | AMES Toxicity: | 0.028 |
| Rat Oral Acute Toxicity: | 0.063 | Maximum Recommended Daily Dose: | 0.032 |
| Skin Sensitization: | 0.955 | Carcinogencity: | 0.054 |
| Eye Corrosion: | 0.996 | Eye Irritation: | 0.952 |
| Respiratory Toxicity: | 0.863 |
——————————————————————————————————————————
*Note: the ADMET properties was calculated by ADMETlab 2.0. Reference: PMID: 33893803.
Similar Compounds*
Compounds similar to EMNPD with top10 similarity:
| Similar NPs | Similar Drugs | ||||||
|---|---|---|---|---|---|---|---|
| NPs ID | NPs 2D Structure | Similarity Score | TTD ID | Drug 2D Structure | Similarity Score | ||
| ENC000380 |  |
0.891 | D00AOJ | 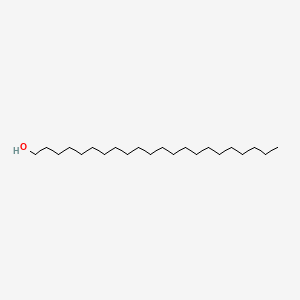 |
0.694 | ||
| ENC000527 |  |
0.833 | D07ILQ | 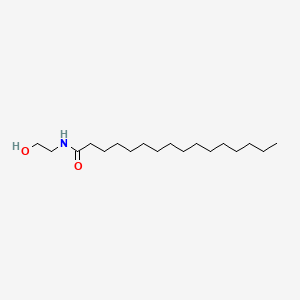 |
0.597 | ||
| ENC000428 | 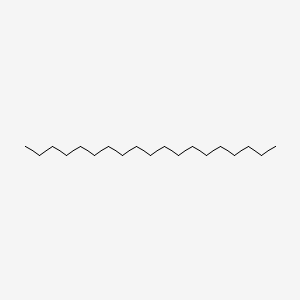 |
0.833 | D00FGR | 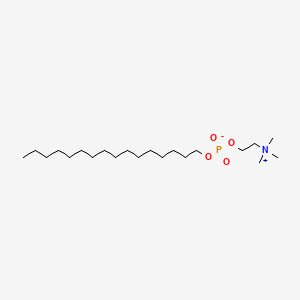 |
0.548 | ||
| ENC000521 |  |
0.833 | D0Z5SM |  |
0.535 | ||
| ENC000284 |  |
0.833 | D05ATI |  |
0.457 | ||
| ENC000745 |  |
0.794 | D0O1PH |  |
0.440 | ||
| ENC000285 |  |
0.794 | D00STJ |  |
0.420 | ||
| ENC000486 |  |
0.783 | D0T9TJ | 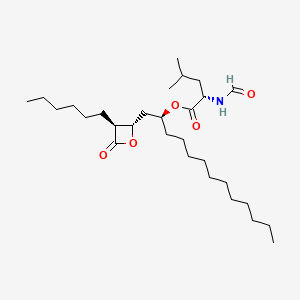 |
0.354 | ||
| ENC000400 | 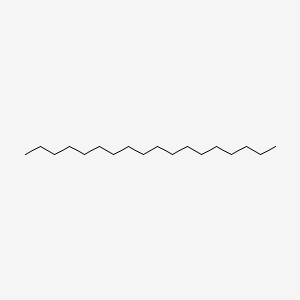 |
0.783 | D0P1RL | 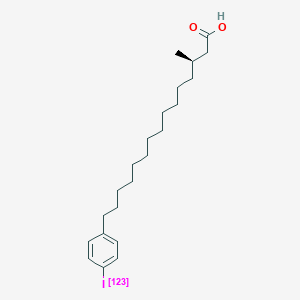 |
0.340 | ||
| ENC001124 | 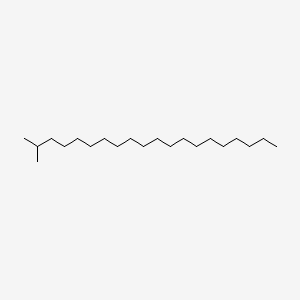 |
0.769 | D00MLW | 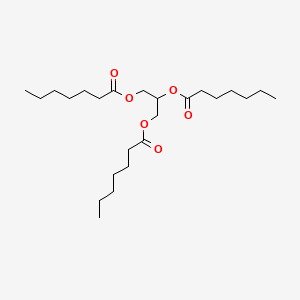 |
0.324 | ||