NPs Basic Information
|
Name |
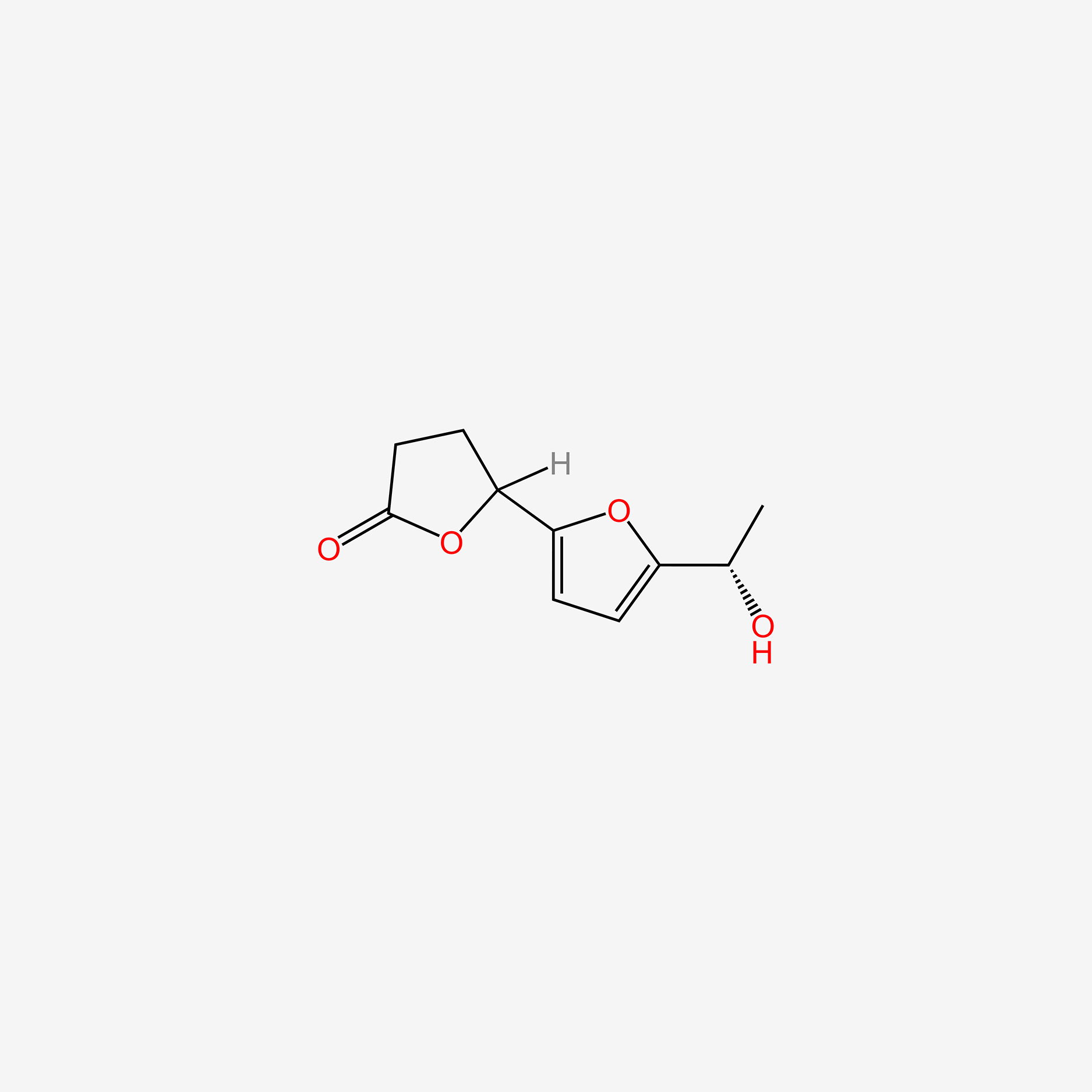
3,4-dihydro-5′-[(1R)-1-hydroxyethyl][2,2′-bifuran]-5(2H)-one
|
| Molecular Formula | C10H12O4 | |
| IUPAC Name* |
5-[5-(1-hydroxyethyl)furan-2-yl]oxolan-2-one
|
|
| SMILES |
CC(O)c1ccc(C2CCC(=O)O2)o1
|
|
| InChI |
InChI=1S/C10H12O4/c1-6(11)7-2-3-8(13-7)9-4-5-10(12)14-9/h2-3,6,9,11H,4-5H2,1H3/t6-,9?/m0/s1
|
|
| InChIKey |
APBQCQCZDOLYQU-AADKRJSRSA-N
|
|
| Synonyms |
NA
|
|
| CAS | NA | |
| PubChem CID | NA | |
| ChEMBL ID | NA |
*Note: the IUPAC Name was calculated by STOUT. Reference: PMID:33906675.
Chemical Classification: |
|
|
|---|
——————————————————————————————————————————
NPs Species Source
| Endophyte ID | Endophyte Name | Family | Genus | Taxonomy ID | GenBank ID | Closest GenBank ID | Reference | |
|---|---|---|---|---|---|---|---|---|
| Endophyte ID | Endophyte Name | Family | Genus | Taxonomy ID | GenBank ID | Closest GenBank ID | Reference |
NPs Biological Activity
| Bioactivity Name | Target ID | Target Name | Target Type | Target Organism | Target Organism ID | Potency of Bioactivity | Activity Type | Value | Unit | Endophyte ID | Endophyte Name | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Bioactivity Name | Target ID | Target Name | Target Type | Target Organism | Target Organism ID | Potency of Bioactivity | Activity Type | Value | Unit | Endophyte ID | Endophyte Name |
NPs Physi-Chem Properties
| Molecular Weight: | 196.2 | ALogp: | 1.7 |
| HBD: | 1 | HBA: | 4 |
| Rotatable Bonds: | 2 | Lipinski's rule of five: | Accepted |
| Polar Surface Area: | 59.7 | Aromatic Rings: | 2 |
| Heavy Atoms: | 14 | QED Weighted: | 0.737 |
——————————————————————————————————————————
NPs ADMET Properties*
ADMET: Absorption
| Caco-2 Permeability: | -4.647 | MDCK Permeability: | 0.00005800 |
| Pgp-inhibitor: | 0.195 | Pgp-substrate: | 0.014 |
| Human Intestinal Absorption (HIA): | 0.011 | 20% Bioavailability (F20%): | 0.004 |
| 30% Bioavailability (F30%): | 0.447 |
——————————————————————————————————————————
ADMET: Distribution
| Blood-Brain-Barrier Penetration (BBB): | 0.331 | Plasma Protein Binding (PPB): | 73.31% |
| Volume Distribution (VD): | 1.619 | Fu: | 39.23% |
——————————————————————————————————————————
ADMET: Metabolism
| CYP1A2-inhibitor: | 0.131 | CYP1A2-substrate: | 0.208 |
| CYP2C19-inhibitor: | 0.183 | CYP2C19-substrate: | 0.389 |
| CYP2C9-inhibitor: | 0.054 | CYP2C9-substrate: | 0.846 |
| CYP2D6-inhibitor: | 0.003 | CYP2D6-substrate: | 0.615 |
| CYP3A4-inhibitor: | 0.024 | CYP3A4-substrate: | 0.352 |
——————————————————————————————————————————
ADMET: Excretion
| Clearance (CL): | 4.572 | Half-life (T1/2): | 0.867 |
——————————————————————————————————————————
ADMET: Toxicity
| hERG Blockers: | 0.011 | Human Hepatotoxicity (H-HT): | 0.523 |
| Drug-inuced Liver Injury (DILI): | 0.409 | AMES Toxicity: | 0.069 |
| Rat Oral Acute Toxicity: | 0.156 | Maximum Recommended Daily Dose: | 0.184 |
| Skin Sensitization: | 0.12 | Carcinogencity: | 0.77 |
| Eye Corrosion: | 0.004 | Eye Irritation: | 0.068 |
| Respiratory Toxicity: | 0.032 |
——————————————————————————————————————————
*Note: the ADMET properties was calculated by ADMETlab 2.0. Reference: PMID: 33893803.
Similar Compounds*
Compounds similar to EMNPD with top10 similarity:
| Similar NPs | Similar Drugs | ||||||
|---|---|---|---|---|---|---|---|
| NPs ID | NPs 2D Structure | Similarity Score | TTD ID | Drug 2D Structure | Similarity Score | ||
| ENC005255 | 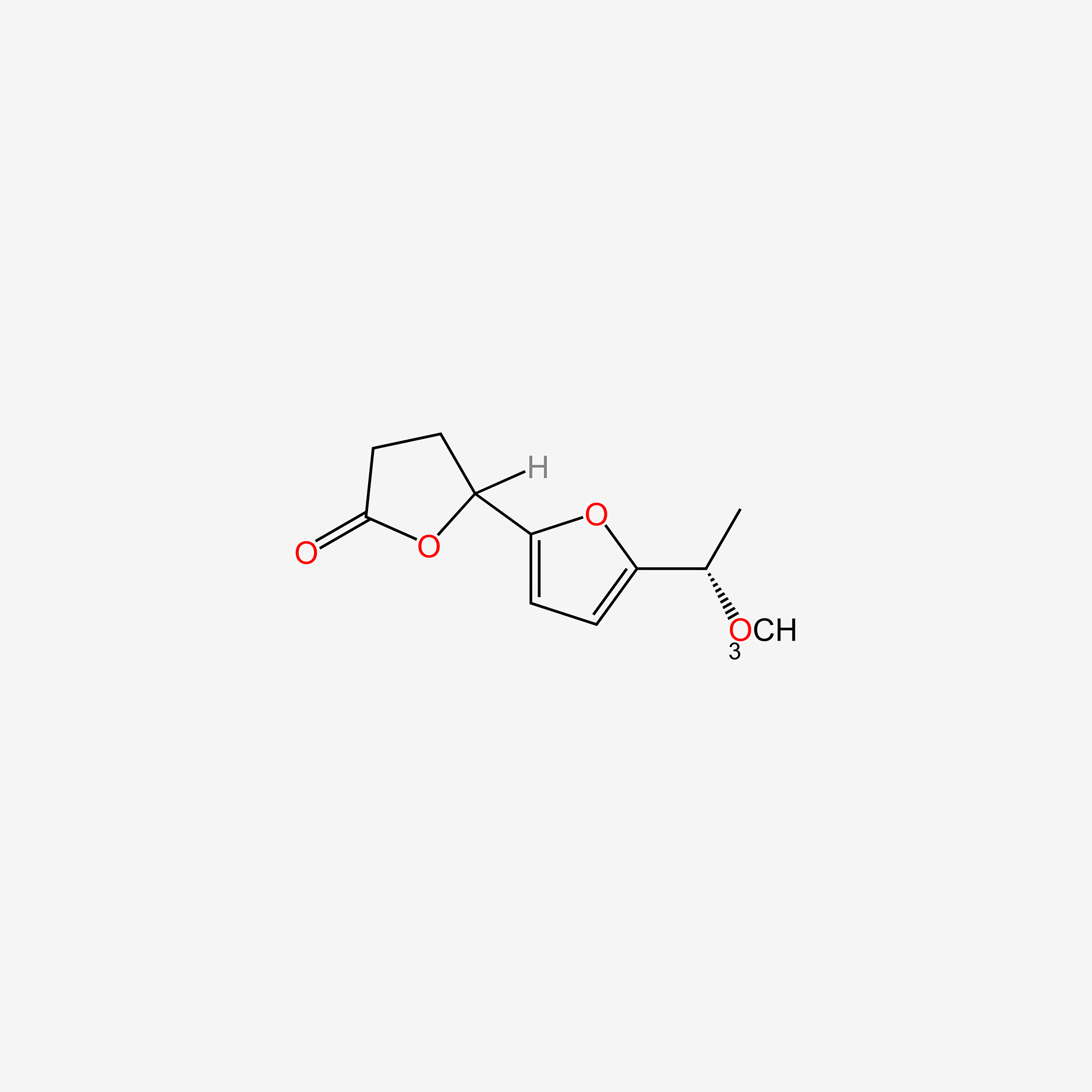 |
0.723 | D04JHN |  |
0.222 | ||
| ENC003462 | 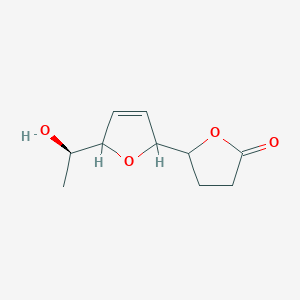 |
0.368 | D02IIW | 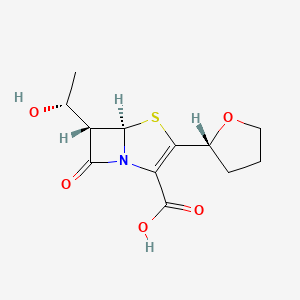 |
0.213 | ||
| ENC003112 | 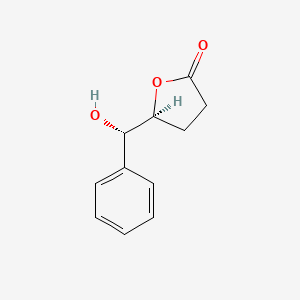 |
0.362 | D0TY5N | 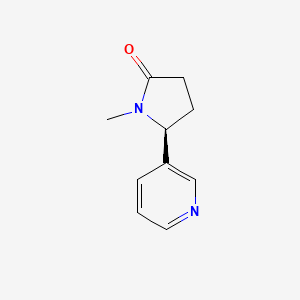 |
0.206 | ||
| ENC004714 | 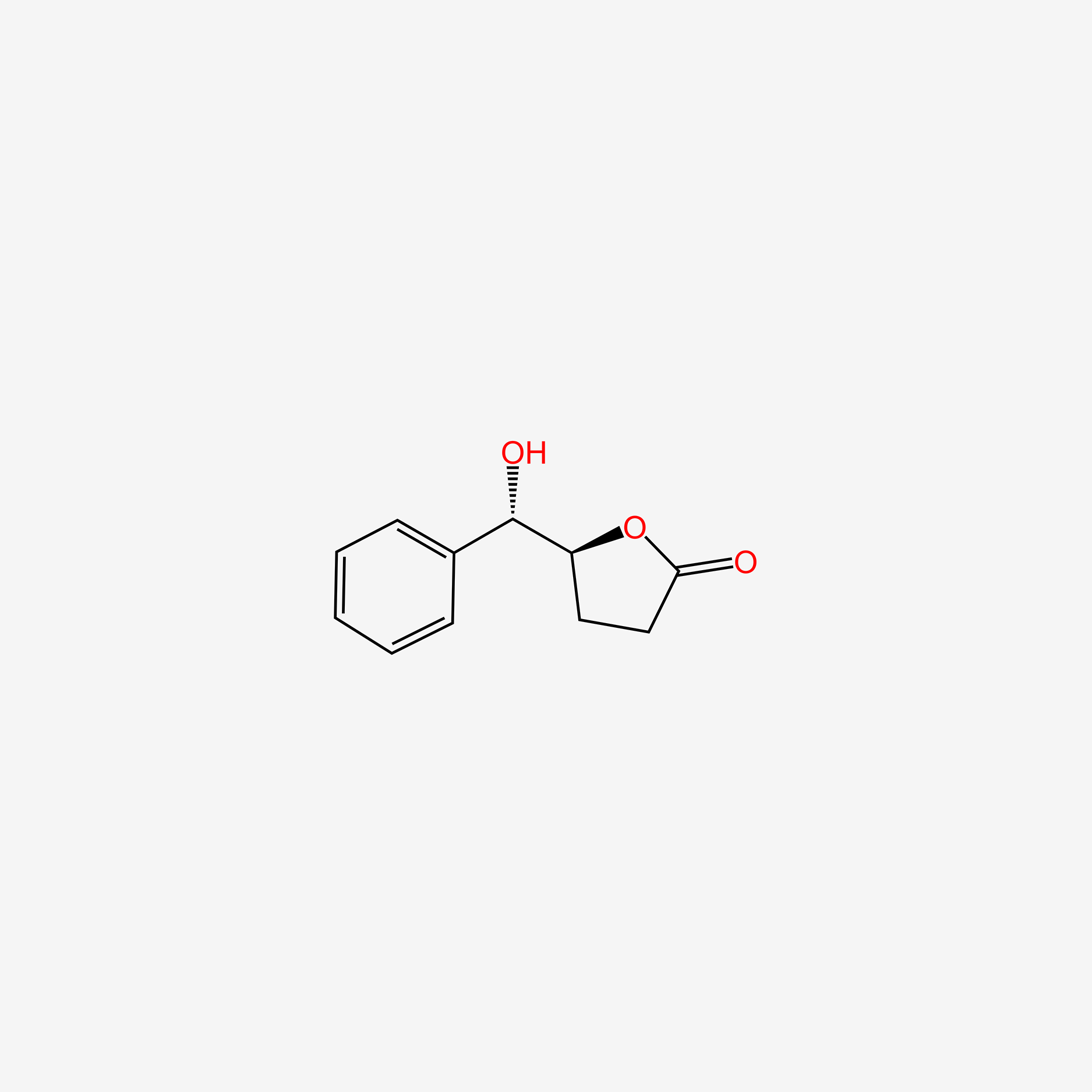 |
0.362 | D02NSF |  |
0.202 | ||
| ENC001329 | 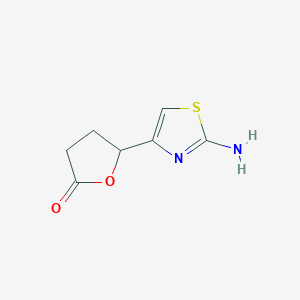 |
0.352 | D00ZFP | 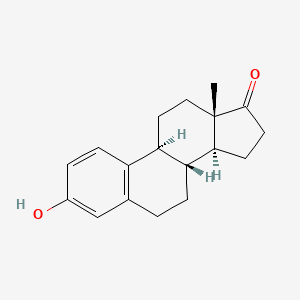 |
0.200 | ||
| ENC000238 | 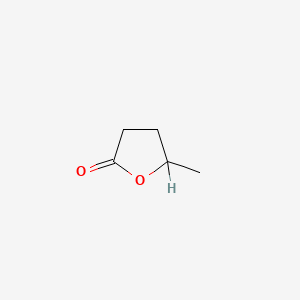 |
0.318 | D0X5KF | 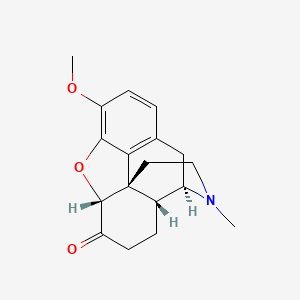 |
0.200 | ||
| ENC005755 | 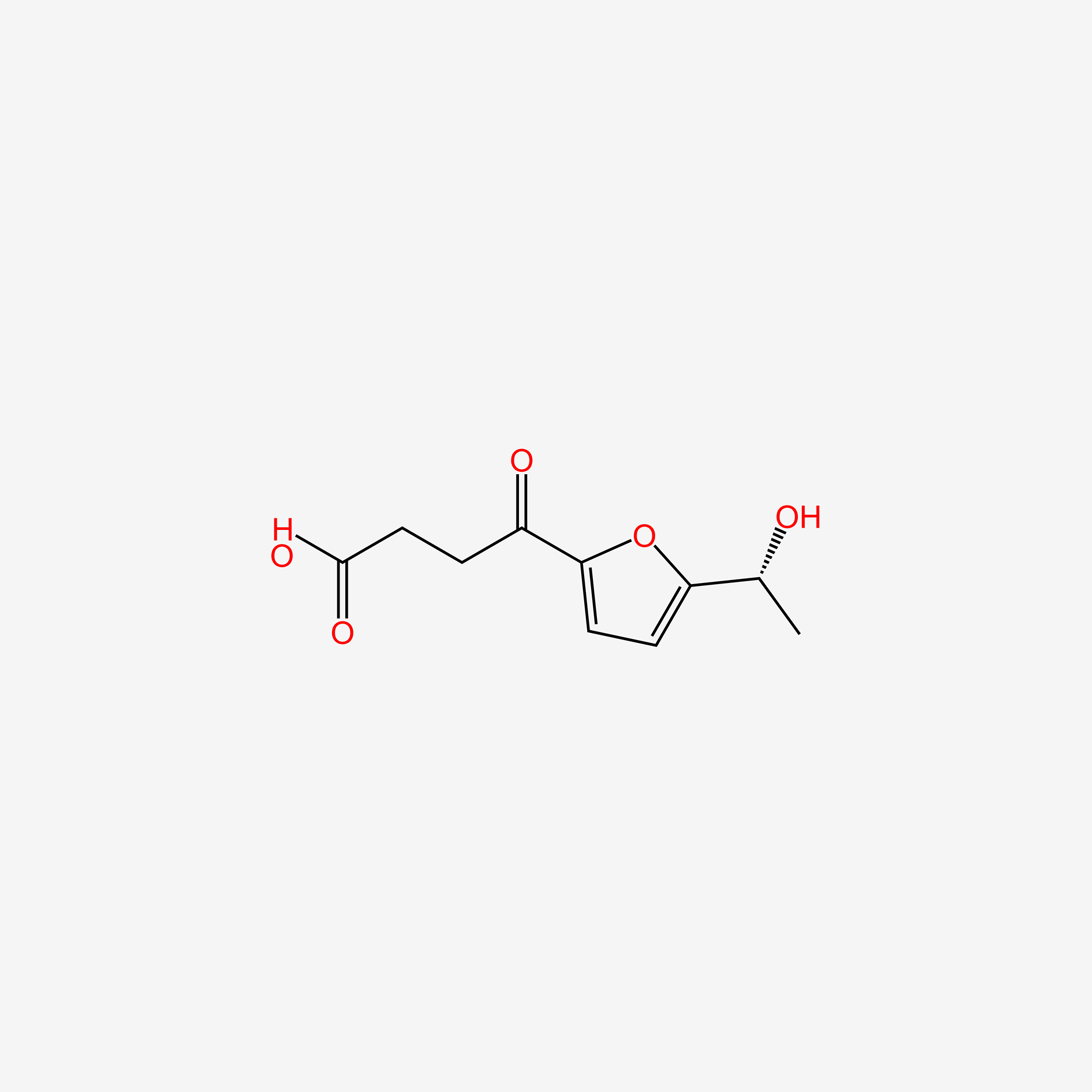 |
0.317 | D03CUF | 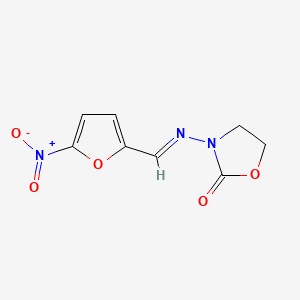 |
0.200 | ||
| ENC005253 | 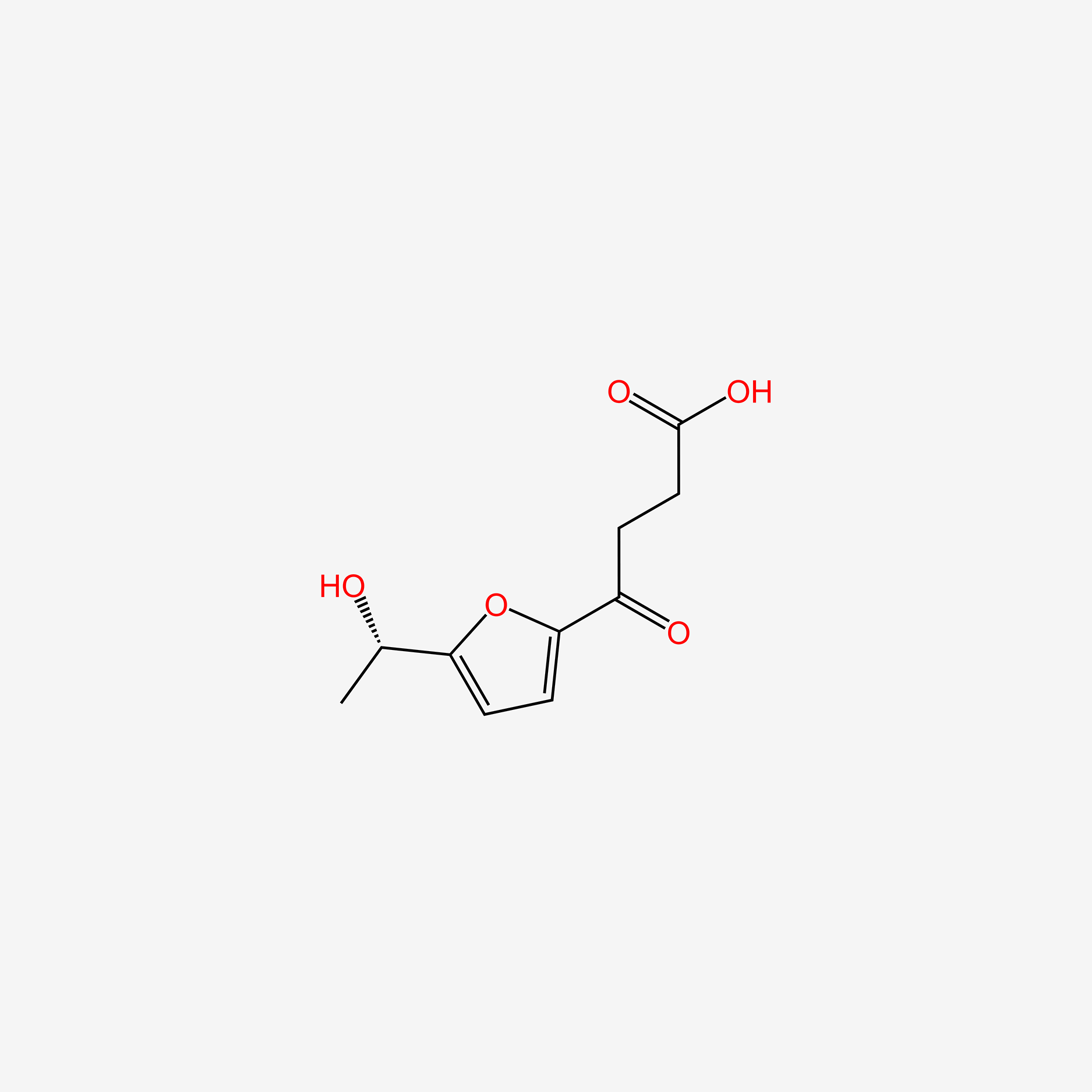 |
0.317 | D09SSC |  |
0.198 | ||
| ENC000899 | 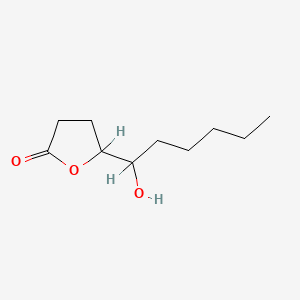 |
0.293 | D03GCJ | 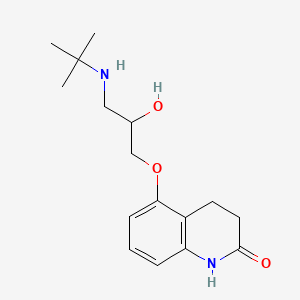 |
0.198 | ||
| ENC003623 | 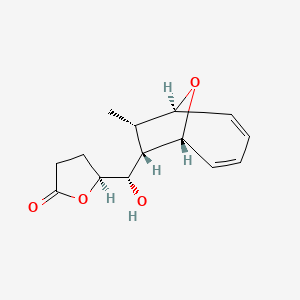 |
0.286 | D00IUG | 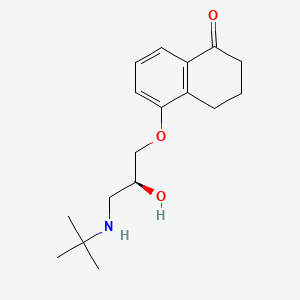 |
0.198 | ||