NPs Basic Information
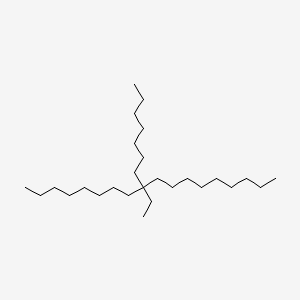
|
Name |
Octadecane, 9-ethyl-9-heptyl-
|
| Molecular Formula | C27H56 | |
| IUPAC Name* |
9-ethyl-9-heptyloctadecane
|
|
| SMILES |
CCCCCCCCCC(CC)(CCCCCCC)CCCCCCCC
|
|
| InChI |
InChI=1S/C27H56/c1-5-9-12-15-17-20-23-26-27(8-4,24-21-18-14-11-7-3)25-22-19-16-13-10-6-2/h5-26H2,1-4H3
|
|
| InChIKey |
LRJHOBBNMAGOIG-UHFFFAOYSA-N
|
|
| Synonyms |
Octadecane, 9-ethyl-9-heptyl-; 9-Ethyl-9-heptyloctadecane; 9-Ethyl-9-n-heptyloctadecane; 55282-27-4; NSC170888; 9-Ethyl-9-heptyloctadecane #; DTXSID70305493; NSC-170888
|
|
| CAS | 55282-27-4 | |
| PubChem CID | 298664 | |
| ChEMBL ID | NA |
*Note: the IUPAC Name was collected from PubChem.
Chemical Classification: |
|
|
|---|
——————————————————————————————————————————
NPs Species Source
| Endophyte ID | Endophyte Name | Family | Genus | Taxonomy ID | GenBank ID | Closest GenBank ID | Reference | |
|---|---|---|---|---|---|---|---|---|
| Endophyte ID | Endophyte Name | Family | Genus | Taxonomy ID | GenBank ID | Closest GenBank ID | Reference |
NPs Biological Activity
| Bioactivity Name | Target ID | Target Name | Target Type | Target Organism | Target Organism ID | Potency of Bioactivity | Activity Type | Value | Unit | Endophyte ID | Endophyte Name | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Bioactivity Name | Target ID | Target Name | Target Type | Target Organism | Target Organism ID | Potency of Bioactivity | Activity Type | Value | Unit | Endophyte ID | Endophyte Name |
NPs Physi-Chem Properties
| Molecular Weight: | 380.7 | ALogp: | 14.4 |
| HBD: | 0 | HBA: | 0 |
| Rotatable Bonds: | 22 | Lipinski's rule of five: | Rejected |
| Polar Surface Area: | 0.0 | Aromatic Rings: | 0 |
| Heavy Atoms: | 27 | QED Weighted: | 0.134 |
——————————————————————————————————————————
NPs ADMET Properties*
ADMET: Absorption
| Caco-2 Permeability: | -5.12 | MDCK Permeability: | 0.00000388 |
| Pgp-inhibitor: | 0 | Pgp-substrate: | 0 |
| Human Intestinal Absorption (HIA): | 0.002 | 20% Bioavailability (F20%): | 0.364 |
| 30% Bioavailability (F30%): | 1 |
——————————————————————————————————————————
ADMET: Distribution
| Blood-Brain-Barrier Penetration (BBB): | 0.008 | Plasma Protein Binding (PPB): | 98.35% |
| Volume Distribution (VD): | 4.033 | Fu: | 1.27% |
——————————————————————————————————————————
ADMET: Metabolism
| CYP1A2-inhibitor: | 0.049 | CYP1A2-substrate: | 0.17 |
| CYP2C19-inhibitor: | 0.12 | CYP2C19-substrate: | 0.435 |
| CYP2C9-inhibitor: | 0.04 | CYP2C9-substrate: | 0.849 |
| CYP2D6-inhibitor: | 0.426 | CYP2D6-substrate: | 0.043 |
| CYP3A4-inhibitor: | 0.354 | CYP3A4-substrate: | 0.04 |
——————————————————————————————————————————
ADMET: Excretion
| Clearance (CL): | 4.254 | Half-life (T1/2): | 0.012 |
——————————————————————————————————————————
ADMET: Toxicity
| hERG Blockers: | 0.401 | Human Hepatotoxicity (H-HT): | 0.052 |
| Drug-inuced Liver Injury (DILI): | 0.041 | AMES Toxicity: | 0.003 |
| Rat Oral Acute Toxicity: | 0.025 | Maximum Recommended Daily Dose: | 0.076 |
| Skin Sensitization: | 0.931 | Carcinogencity: | 0.022 |
| Eye Corrosion: | 0.985 | Eye Irritation: | 0.914 |
| Respiratory Toxicity: | 0.177 |
——————————————————————————————————————————
*Note: the ADMET properties was calculated by ADMETlab 2.0. Reference: PMID: 33893803.
Similar Compounds*
Compounds similar to EMNPD with top10 similarity:
| Similar NPs | Similar Drugs | ||||||
|---|---|---|---|---|---|---|---|
| NPs ID | NPs 2D Structure | Similarity Score | TTD ID | Drug 2D Structure | Similarity Score | ||
| ENC000428 | 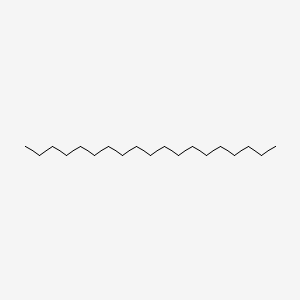 |
0.630 | D00AOJ | 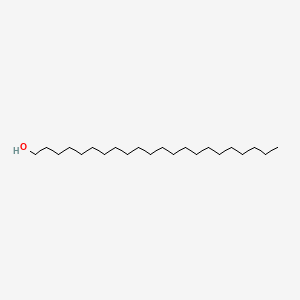 |
0.532 | ||
| ENC001034 | 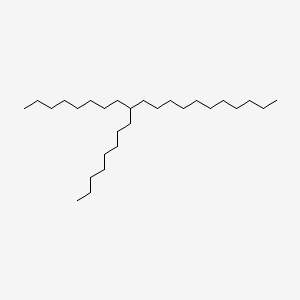 |
0.629 | D00FGR | 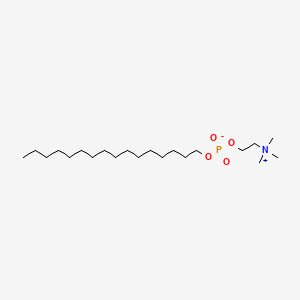 |
0.448 | ||
| ENC000285 | 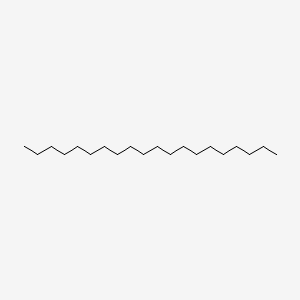 |
0.627 | D07ILQ | 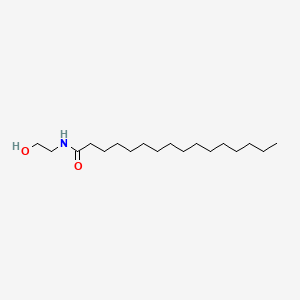 |
0.442 | ||
| ENC000430 | 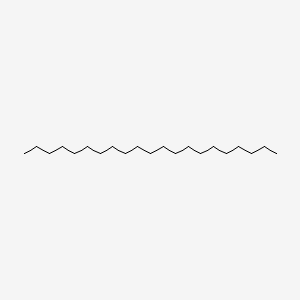 |
0.624 | D0Z5SM |  |
0.409 | ||
| ENC000432 |  |
0.621 | D00MLW |  |
0.376 | ||
| ENC000442 |  |
0.618 | D0T9TJ |  |
0.367 | ||
| ENC000446 |  |
0.615 | D00STJ | 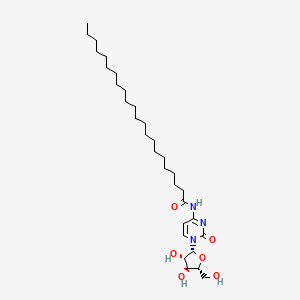 |
0.355 | ||
| ENC000400 | 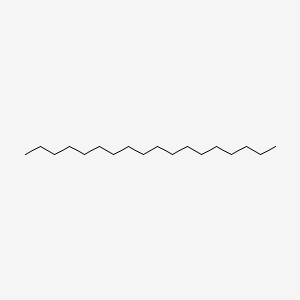 |
0.613 | D0O1PH | 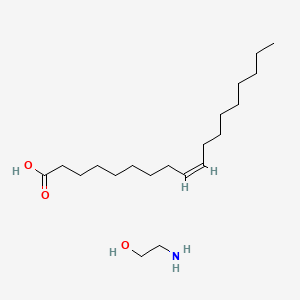 |
0.349 | ||
| ENC005691 | 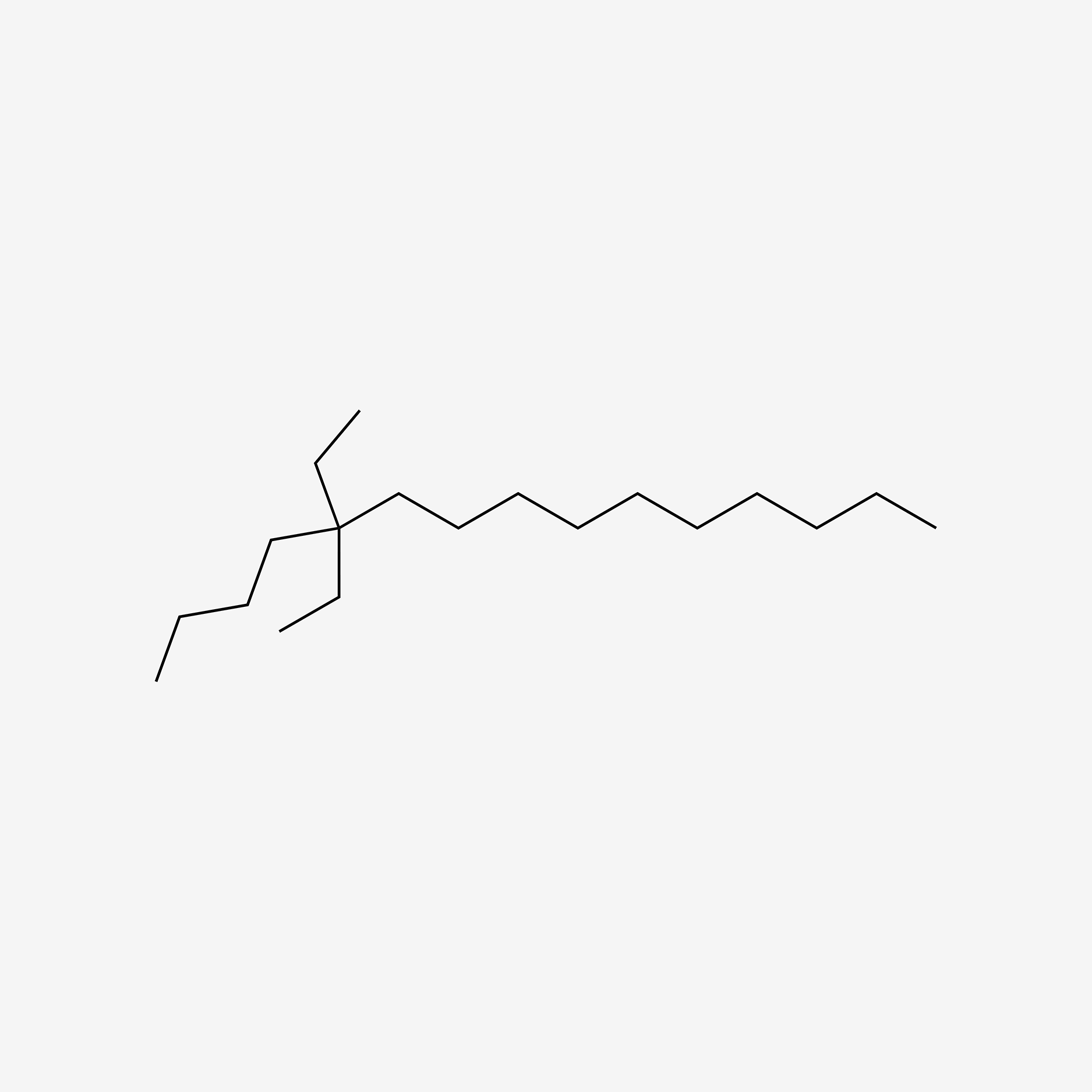 |
0.605 | D05ATI |  |
0.348 | ||
| ENC001180 |  |
0.604 | D0Z1QC |  |
0.342 | ||